Buy Solriamfetol (Sunosi) Cas 178429-62-4
Buy Solriamfetol (Sunosi) Cas 178429-62-4
Solriamfetol, sold under the brand name Sunosi, is a wakefulness-promoting medication used in the treatment of excessive sleepiness related to narcolepsy and sleep apnea.[1][5][6] It is taken by mouth.[1]
Common side effects of solriamfetol include headache, nausea, anxiety, and trouble sleeping.[1] It is a norepinephrine–dopamine reuptake inhibitor (NDRI) and is thought to work by increasing levels of the neurotransmitters norepinephrine and dopamine in the brain.[1][5] Solriamfetol has also been found to act as a TAAR1 agonist, an action that may also be involved in its effects.[7]
The drug was discovered by a subsidiary of SK Group, which licensed rights outside of eleven countries in Asia to Aerial Pharma in 2011.[8] In addition to its approved indication of excessive sleepiness, solriamfetol is under development for certain other uses including the treatment of attention deficit hyperactivity disorder (ADHD), binge eating disorder, and circadian rhythm sleep disorders.[9] The drug is also a potential designer drug.[10]
Medical uses
Solriamfetol is used to promote wakefulness in the treatment of excessive daytime sleepiness associated with narcolepsy or obstructive sleep apnea in adults.[1] It appears to be more effective in improving excessive daytime sleepiness associated with obstructive sleep apnea than certain other wakefulness-promoting agents including modafinil, armodafinil, and pitolisant.[11]
Available forms
Solriamfetol is available in the form of 75 and 150 mg oral tablets.[1]
Side effects
Side effects of solriamfetol include headache, nausea, decreased appetite, insomnia, anxiety, irritability, feeling jittery, dizziness, chest discomfort, heart palpitations, dry mouth, increased sweating, abdominal pain, constipation, and diarrhea.[1]
Misuse potential
Solriamfetol at higher-than-approved doses—specifically doses of 300, 600, and 1,200 mg, which are 2 to 8 times the maximum recommended dose—produces drug-liking responses, including elevated mood and feelings of relaxation, that are similar in degree to those of phentermine (a Schedule IV controlled substance).[1] Elevated mood occurred in 2.4% with placebo, 8 to 24% with solriamfetol, and 10 to 18% with phentermine, while feelings of relaxation occurred in 5% with placebo, 5 to 19% with solriamfetol, and 15 to 20% with phentermine.[1] As such, solriamfetol has significant misuse potential and is a controlled substance in the United States.[1] However, solriamfetol showed less misuse potential than Schedule II controlled stimulants like amphetamine and cocaine.[12] Consequently, the misuse potential of solriamfetol was rated as low and it was placed in the Schedule IV controlled substance category alongside phentermine.[12]Buy Solriamfetol (Sunosi) Cas 178429-62-4
Pharmacology
Pharmacodynamics
Solriamfetol is a norepinephrine–dopamine reuptake inhibitor (NDRI).[1] It binds to the dopamine transporter (DAT) and the norepinephrine transporter (NET) with affinities (Ki) of 14.2 μM and 3.7 μM, respectively.[1] It inhibits the reuptake of dopamine and norepinephrine with IC50 values of 2.9 μM and 4.4 μM, respectively.[1] It has weak affinity for the serotonin transporter (Ki = 81.5 μM) and does not appreciably inhibit serotonin reuptake (IC50 > 100 μM).[1] In addition to its dopamine and norepinephrine reuptake inhibition, solriamfetol has been found to act as an agonist of the human and rodent TAAR1 (EC50 = 10–16 μM) at clinically relevant concentrations similar to those of its DAT and NET inhibition.[7] Solriamfetol has no appreciable affinity for a variety of other targets, including Buy Solriamfetol (Sunosi) Cas 178429-62-4 the dopamine, serotonin, adrenergic, GABA, adenosine, histamine, orexin, benzodiazepine, and acetylcholine receptors.[1]
Pharmacokinetics
The oral bioavailability of solriamfetol is approximately 95%.[1] The median time to peak levels of solriamfetol is 2 hours, with a range of 1.25 to 3.0 hours.[1] A high-fat meal has minimal influence on the peak and total concentrations of solriamfetol, but does delay time to peak levels by approximately 1 hour.[1] The apparent volume of distribution of solriamfetol is approximately 199 L.[1] The plasma protein binding of solriamfetol is 13.3% to 19.4% over a concentration range of 0.059 to 10.1 μg/mL.[1] Solriamfetol is minimally metabolized in humans.[1] It shows first-order elimination with oral administration and has an elimination half-life of about 7.1 hours.[1] The half-life of solriamfetol increases in the context of renal impairment.[1] Approximately 95% of a dose of solriamfetol is eliminated in urine as unchanged solriamfetol and 1% or less is eliminated as the minor inactive metabolite N-acetylsolriamfetol. Buy Solriamfetol (Sunosi) Cas 178429-62-4
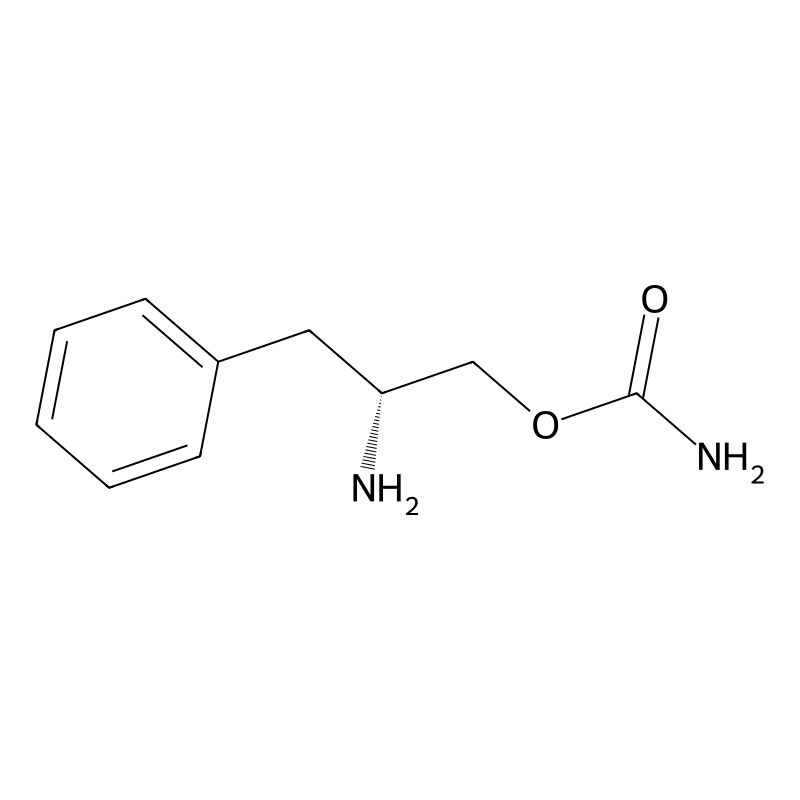
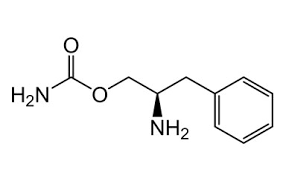
Chemistry
Solriamfetol is a substituted phenethylamine derived from d-phenylalanine and D-phenylalaninol.[13] Its chemical name is (R)-2-amino-3-phenylpropylcarbamate.[14][13] It is also known as O-carbamoyl-D-phenylalaninol. Buy Solriamfetol (Sunosi) Cas 178429-62-4
History
The drug was discovered by a subsidiary of SK Group, which licensed rights outside of eleven countries in Asia to Aerial Pharma in 2011.[8] Aerial ran two Phase II trials of the drug in narcolepsy[15] before selling the license to solriamfetol to Jazz in 2014; Jazz Pharmaceuticals paid Aerial $125 million up front and agreed to pay Aerial and SK up to $272 million in milestone payments, as well as double-digit royalties to SK.[8][16]
In 2019, solriamfetol was approved by the U.S. Food and Drug Administration (FDA) to improve wakefulness in adults with narcolepsy or obstructive sleep apnea.[17][18] It was granted orphan drug designation.[19]
Solriamfetol was approved for medical use in the European Union in January 2020.[4]
In March 2022, it was announced that Axsome Therapeutics would be acquiring Solriamfetol, under the brand name Sunosi, from Jazz Pharmaceuticals, for an upfront sum of $53 million. Jazz will receive a high single-digit royalty on Axsome’s U.S. net sales of Sunosi in the current indication, and a mid-single-digit royalty in the future indications. Axsome will also assume the commitments of Jazz to SK Biopharmaceuticals and Aerial Biopharma.[20]
Society and culture
Names
During development it has been called SKL-N05, ADX-N05, ARL-N05, and JZP-110.[9]
Legal status
In the United States, solriamfetol is a Schedule IV controlled substance,[1] meaning that it has an accepted medical use and a low potential for abuse, but that abuse may lead to physical or psychological dependence.[21] A prescription is required, and can only be refilled up to five times in a six-month period.[22] In countries of the European Union, a prescription is required. Buy Solriamfetol (Sunosi) Cas 178429-62-4
Research
Solriamfetol is under development for the treatment of attention deficit hyperactivity disorder (ADHD), binge eating disorder, and circadian rhythm sleep disorders.[9][23] As of September 2023, it is in phase 3 clinical trials for ADHD and phase 2 clinical trials for binge eating disorder and circadian rhythm sleep disorders.[9][23] A case report of solriamfetol for the treatment of ADHD has been published.[24] Solriamfetol was also under development for the treatment of depressive disorders, but development for this indication was discontinued.[9] In May 2024, the National Institutes of Health (NIH) announced a trial of solriamfetol for the treatment of long COVID.[25] In September 2025, a small double-blind, placebo-controlled study found positive results in “improving fatigue and executive functioning in patients with ME/CFS
Solriamfetol HCl (YKP-10A; ADXN-05; R228060; SKL-N-05; JZP-11; Sunosi), the hydrochloride salt of Solriamfetol which is a wake-promoting agent, is a norepinephrine–dopamine inhibitor that was approved in 2019 for the treatment of excessive sleepiness associated with narcolepsy and sleep apnea.
| Targets |
Dopamine transporter (DAT) and norepinephrine transporter (NET) [1]
– Dopamine transporter (DAT) (IC₅₀ = 0.24 μM) – Norepinephrine transporter (NET) (IC₅₀ = 0.31 μM) – Serotonin transporter (SERT) (IC₅₀ = 1.8 μM) [1] |
|---|---|
| ln Vitro |
– Transporter Inhibition: Solriamfetol demonstrated selective inhibition of DAT and NET in radioligand binding assays, with higher potency against DAT compared to SERT. The compound showed competitive binding to the substrate-binding sites of these transporters, reducing neurotransmitter reuptake [1]
– Receptor Selectivity: No significant binding was observed to adrenergic, dopaminergic, or serotonergic receptors at concentrations up to 10 μM, confirming specificity for transporters [1] |
| ln Vivo |
Solriamfetol was well tolerated, with no significant effect on mean heart rate (+3.7 vs +2.2 bpm, P = .5609), systolic blood pressure (+2.4 vs +1.5 mm Hg, P = .6474), or diastolic blood pressure (+1.1 vs +1.5 mm Hg, P = .8117). There was no statistically significant treatment effect on occurrence of adverse events. Compared to individuals on placebo, individuals on solriamfetol treatment experienced adverse events at a rate of at least 10 percentage points higher in the categories of decreased appetite, headache, gastrointestinal, insomnia, increased energy, cardiovascular, and neurologic. Compared to individuals on placebo, by study endpoint, a greater proportion of individuals in the treatment group met the a priori-defined treatment response (CGI score indicating much or very much improved and AISRS score reduced ≥ 25%: 45% vs 6.9%, P = .0020); those treated with solriamfetol also had greater improvement in total AISRS scores by week 3 through week 6 (P = .0012; week 6 effect size = 1.09). Significantly more solriamfetol-treated adults than placebo-treated adults had 0.5-standard deviation improvement in T-score on the BRIEF-A Global Executive Composite (P = .0173); those treated with solriamfetol also had greater mean change in GAF score (-4.8 vs -0.3, P = .0006) and greater mean MASRS total score change (P = .0047; effect size = 1.23). Mean ESS score improved more with solriamfetol than with placebo (P = .0056), but this difference did not predict AISRS response (P = .3735). There was no significant association between solriamfetol and change in PSQI scores. Conclusions: Solriamfetol may be a novel and effective treatment for the management of ADHD in adults. Further replication in larger trials is indicated. [1]
1. Solriamfetol (150 mg/day) significantly reduced ADHD-RS total score by 14.3±3.2 points vs placebo (7.1±2.8 points; P=0.021) after 6 weeks [1] – ADHD Symptom Improvement: In a 4-week double-blind placebo-controlled trial (N=60), adult ADHD patients treated with solriamfetol (150–300 mg/day) showed significant reductions in ADHD Rating Scale-IV (ADHD-RS-IV) total scores compared to placebo (mean change: -12.3 vs. -5.1; p<0.001). Improvements were sustained across all subscales (inattention, hyperactivity/impulsivity) [1] |
| Enzyme Assay |
– Transporter Binding Assay: Membrane preparations from HEK293 cells expressing human DAT, NET, or SERT were incubated with radiolabeled substrates (³H-DA, ³H-NE, ³H-5-HT) and increasing concentrations of solriamfetol (0.01–10 μM). Non-specific binding was determined using nomifensine (10 μM for DAT/NET) or citalopram (10 μM for SERT). IC₅₀ values were calculated by nonlinear regression analysis [1]
|
| Cell Assay |
– Neurotransmitter Uptake Inhibition: SH-SY5Y cells transfected with DAT or NET were treated with solriamfetol (0.1–10 μM) for 30 minutes, followed by incubation with ³H-DA or ³H-NE. Uptake was terminated by washing, and radioactivity was measured by liquid scintillation counting. Solriamfetol dose-dependently reduced DA and NE uptake with EC₅₀ values of 0.32 μM and 0.45 μM, respectively [1]
– Cell Viability: No significant cytotoxicity was observed in neuronal cells exposed to solriamfetol up to 100 μM, as assessed by MTT assay [1] |
| Animal Protocol |
Sixty adults with DSM-5 ADHD participated from August 2021 through January 2023 in a remotely conducted, randomized, double-blind, placebo-controlled, 6-week dose-optimization trial of 75 mg or 150 mg of solriamfetol. Measures included the Adult ADHD Investigator Symptom Rating Scale (AISRS), which was our primary outcome measure, as well as the Clinical Global Impressions scale (CGI), vital signs, the Global Assessment of Functioning (GAF), the Behavior Rating Inventory of Executive Function-Adult Form (BRIEF-A), the Epworth Sleepiness Scale (ESS), the Pittsburgh Sleep Quality Index (PSQI), and a modified Adult ADHD Self-Report Scale (MASRS).
– Pharmacokinetic Study in Rats: Male Sprague-Dawley rats (n=6/group) received single oral doses of solriamfetol (10–50 mg/kg). Blood samples were collected at predefined time points, and plasma concentrations were analyzed by LC-MS/MS. The compound exhibited linear pharmacokinetics with a Tₘₐₓ of 1.5–2 hours and a terminal half-life of 12–14 hours [1] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
Oral bioavailability of solriamfetol is approximately 95%. Peak plasma concentration is reached in 2 hours (with a range of 1.25 to 3 hours) in fasted patients. When solriamfetol is taken with a high fat meal, the time to peak plasma concentration increases to 3 hours. 95% of solriamfetol is recovered in urine unchanged by metabolism. Less than 1% of solriamfetol is recovered as N-acetyl solriamfetol. 199L. Other studies have found the volume of distribution to be 158.2L ± 37.3L in fasted subjects and 159.8L ± 38.9L in fed subjects. Renal clearance is 18.2L/h and total clearance is 19.5L/h. Other studies have found clearance to be 18.4 ± 4.2L/h in fasted subjects and 18.8 ± 4.2L/h in fed subjects. Metabolism / Metabolites Biological Half-Life 1. Median Tmax: 2.0 h (range 1.5–3.5 h) post-dose [1] – Human Pharmacokinetics: Following oral administration, solriamfetol is rapidly absorbed (Tₘₐₓ = 1.5–2 hours) with an absolute bioavailability of 82%. The drug is primarily metabolized by CYP2C19 and CYP3A4, with a mean elimination half-life of 12.5 hours. Plasma protein binding is approximately 65% [1] |
| Toxicity/Toxicokinetics |
Hepatotoxicity
In placebo-controlled trials of solriamfetol in patients with narcolepsy, minor serum aminotransferase elevations occurred in a small proportion of patients during therapy, but the rates of enzyme elevations overall were similar to those in placebo recipients. In preregistration trials, there were no instances of clinically apparent liver injury or serum aminotransferase elevations with jaundice attributable to solriamfetol. Since its approval in 2019, there have been no publications describing clinically apparent liver injury due to solriamfetol. Likelihood score: E (unlikely cause of acute liver injury with jaundice). Effects During Pregnancy and Lactation Protein Binding 1. Treatment-emergent adverse events (TEAEs): Headache (24.1% vs 10.3% placebo), nausea (17.2% vs 3.4%), insomnia (13.8% vs 0%) [1] – Acute Toxicity: The LD₅₀ of solriamfetol in mice and rats exceeded 2000 mg/kg (oral). No significant organ toxicity was observed in 28-day repeat-dose studies at doses up to 300 mg/kg/day [1] |
| References |
[1]. Solriamfetol for Attention-Deficit/Hyperactivity Disorder in Adults: A Double-Blind Placebo-Controlled Pilot Study. J Clin Psychiatry. 2023 Oct 9;84(6):23m14934.
|
| Additional Infomation |
Solriamfetol is a DEA Schedule IV controlled substance. Substances in the DEA Schedule IV have a low potential for abuse relative to substances in Schedule III. It is a Stimulants substance.
Solriamfetol marketed under the brand name Sunosi by Jazz Pharmaceuticals in the United States is a dopamine and norepinephrine reuptake inhibitor (DNRI) indicated in treating daytime sleepiness associated with narcolepsy or obstructive sleep apnea. Solriamfetol was given FDA approval in 2019. Solriamfetol is a Dopamine and Norepinephrine Reuptake Inhibitor. The mechanism of action of solriamfetol is as a Dopamine Uptake Inhibitor, and Norepinephrine Uptake Inhibitor. Solriamfetol is dopamine and norepinephrine reuptake inhibitor that is used in the therapy of excessive daytime sleepiness and cataplexy in patients with narcolepsy. Solriamfetol has not been associated with serum enzyme elevations during therapy or to instances of idiosyncratic acute liver injury. See also: Solriamfetol Hydrochloride (is active moiety of). Drug Indication Mechanism of Action Pharmacodynamics 1. Study design: Randomized, double-blind, placebo-controlled trial in 29 adults with ADHD (DSM-5 criteria) [1] – Mechanism of Action: Solriamfetol enhances dopaminergic and noradrenergic neurotransmission by blocking DAT and NET, thereby increasing synaptic concentrations of dopamine and norepinephrine. This dual action targets both inattention and hyperactivity symptoms of ADHD [1] |
Solriamfetol
Content Navigation
CAS Number
Product Name
IUPAC Name
Molecular Formula
Molecular Weight
InChI
InChI Key
SMILES
solubility
Synonyms
Canonical SMILES
Isomeric SMILES
Understanding the Mechanism of Action in Narcolepsy
Narcolepsy is characterized by excessive daytime sleepiness (EDS) and cataplexy, a sudden loss of muscle control triggered by emotions. The exact mechanism by which Solriamfetol improves wakefulness in narcolepsy is still under investigation. However, research suggests it might increase the levels of dopamine and norepinephrine in the brain, promoting wakefulness and alertness []. Studies are ongoing to further elucidate the specific neural pathways involved in Solriamfetol’s action within the context of narcolepsy [].
Potential Benefits in Other Sleep-Wake Disorders
Given its wakefulness-promoting effects in narcolepsy, researchers are exploring the use of Solriamfetol in other sleep disorders characterized by EDS. These include idiopathic hypersomnia, a condition of excessive daytime sleepiness without cataplexy, and shift work sleep disorder, which affects individuals who work irregular hours [, ]. Studies are investigating Solriamfetol’s efficacy and safety in improving alertness and reducing sleepiness in these patient populations [, ].
Applications Beyond Sleep Disorders
Solriamfetol’s effects on dopamine and norepinephrine reuptake have led researchers to explore its potential benefits in neurological conditions beyond sleep disorders. Parkinson’s disease, a neurodegenerative disorder characterized by tremors and movement difficulties, is one area of investigation. Dopamine deficiency plays a role in Parkinson’s disease, and Solriamfetol’s ability to increase dopamine levels might offer therapeutic potential []. However, further research is needed to determine its efficacy and safety in managing Parkinson’s disease symptoms [].
Exploring Cognitive Enhancement
Solriamfetol’s impact on alertness and wakefulness has also sparked interest in its potential cognitive-enhancing properties. Some studies suggest that Solriamfetol may improve cognitive function in healthy individuals []. However, more research is required to confirm these findings and understand the long-term effects of Solriamfetol on cognition in both healthy and patient populations [].
Solriamfetol is a pharmacological compound primarily used as a medication to enhance wakefulness in adults suffering from excessive daytime sleepiness associated with narcolepsy or obstructive sleep apnea. It is marketed under the brand name SUNOSI. The chemical structure of solriamfetol is characterized by its molecular formula C₁₀H₁₄N₂O₂, and it is classified as a selective dopamine and norepinephrine reuptake inhibitor. The compound was developed to target neurotransmitter systems involved in sleep-wake regulation, specifically enhancing the availability of dopamine and norepinephrine in the brain .
Solriamfetol acts as a dopamine and norepinephrine reuptake inhibitor (DNRI). This means it prevents the reabsorption of these neurotransmitters by presynaptic neurons, leading to increased levels in the synapse. Dopamine and norepinephrine play a crucial role in wakefulness and alertness, and by enhancing their activity, Solriamfetol helps improve daytime wakefulness in individuals with EDS [, ].
The biological activity of solriamfetol is centered on its role as a stimulant. Clinical studies have shown that it effectively reduces excessive daytime sleepiness and improves wakefulness in patients with narcolepsy and obstructive sleep apnea. In phase III clinical trials, doses ranging from 37.5 mg to 150 mg demonstrated significant efficacy compared to placebo over a treatment period of up to 52 weeks . Common side effects include headache, decreased appetite, nausea, anxiety, and insomnia .
Solriamfetol is primarily indicated for the treatment of excessive daytime sleepiness associated with narcolepsy and obstructive sleep apnea. Its approval by regulatory bodies such as the U.S. Food and Drug Administration underscores its therapeutic utility in managing conditions that impair wakefulness. Additionally, ongoing research may explore its potential in other areas related to mood disorders or cognitive enhancement due to its dopaminergic activity .
Several compounds share similarities with solriamfetol in their pharmacological profiles or mechanisms of action:
| Compound Name | Mechanism of Action | Unique Features |
|---|---|---|
| Modafinil | Dopamine reuptake inhibition | Less potent than solriamfetol in norepinephrine inhibition |
| Armodafinil | Dopamine reuptake inhibition | Longer half-life compared to modafinil |
| Amphetamine | Release of dopamine and norepinephrine | More potent stimulant effects; higher abuse potential |
| Methylphenidate | Dopamine reuptake inhibition | Primarily used for ADHD; different pharmacokinetics |
Uniqueness of Solriamfetol: Unlike many traditional stimulants such as amphetamines, solriamfetol offers a more targeted approach by selectively inhibiting dopamine and norepinephrine reuptake without significant serotonin involvement. This specificity may result in a different side effect profile and lower potential for abuse compared to more potent stimulants .
Key Synthetic Routes for Solriamfetol Production
The manufacture of solriamfetol, a dopamine and norepinephrine reuptake inhibitor approved for treating excessive daytime sleepiness, involves several well-characterized synthetic approaches that have evolved from laboratory-scale preparations to large-scale manufacturing processes [1] [2]. The compound’s relatively simple structure, containing a single chiral center, has enabled the development of efficient synthetic routes that maintain high stereochemical purity while achieving commercially viable yields.
D-Phenylalanine Methyl Ester as a Precursor
The most widely documented synthetic pathway for solriamfetol utilizes D-phenylalanine methyl ester as the primary starting material, representing a chiral pool approach that ensures the desired R-configuration in the final product [1] [3] . This route capitalizes on the commercial availability of D-phenylalanine methyl ester hydrochloride, which eliminates the need for complex chiral resolution steps during the synthesis process.
The synthetic sequence begins with the dissociation of the hydrochloride salt of D-phenylalanine methyl ester, followed by reduction of the ester functionality to yield the corresponding amino alcohol intermediate [1]. This reduction step typically employs sodium borohydride or lithium aluminum hydride under controlled conditions to prevent over-reduction or unwanted side reactions. The resulting D-phenylalaninol serves as the immediate precursor for carbamate formation, the final step in solriamfetol synthesis.
The carbamate formation represents the most critical transformation in this synthetic route, where D-phenylalaninol is treated with sodium cyanate under acidic conditions to introduce the carbamate functionality [1] [5]. This reaction requires careful pH control and temperature management to ensure optimal yield while minimizing the formation of process-related impurities. The acidic conditions facilitate the nucleophilic attack of the primary alcohol on the electrophilic carbon of the cyanate, leading to the formation of the desired carbamate bond.
Studies have demonstrated that this pathway can achieve yields exceeding 89% for the carbamate formation step when optimized conditions are employed [1]. The high efficiency of this transformation, combined with the readily available starting material, makes this route particularly attractive for large-scale manufacturing applications where cost-effectiveness and process reliability are paramount considerations.
Novel Intermediates in Large-Scale Synthesis
Recent patent disclosures have revealed innovative approaches to solriamfetol synthesis that incorporate novel intermediates designed specifically for large-scale manufacturing requirements [5] [6] [3]. These advanced synthetic strategies address the limitations of traditional laboratory-scale procedures by introducing process improvements that enhance both yield and purity while reducing manufacturing costs.
One particularly noteworthy development involves the use of protected amino alcohol intermediates that allow for more selective carbamate formation reactions [6]. In this approach, D-phenylalaninol is first treated with protecting groups that temporarily mask the amino functionality, preventing unwanted side reactions during subsequent transformations. The protected intermediate then undergoes carbamate formation under milder conditions, followed by deprotection to yield the final product. This strategy has demonstrated superior impurity profiles compared to direct carbamate formation methods.
Another innovative approach disclosed in patent WO2020035769A1 employs tartaric acid resolution techniques to obtain high-purity D-phenylalanine derivatives [3]. This method involves the formation of diastereomeric tartrate salts that can be separated through selective crystallization, providing access to optically pure starting materials that subsequently undergo efficient conversion to solriamfetol. The tartaric acid resolution approach is particularly valuable when starting from racemic precursors, as it enables the isolation of the desired R-enantiomer with high optical purity.
The patent literature also describes novel synthetic intermediates that incorporate benzyl carbamate protecting groups [7]. These intermediates offer enhanced stability during processing and storage while maintaining high reactivity toward the final carbamate formation step. The benzyl protecting group can be removed under mild hydrogenation conditions, providing a clean conversion to solriamfetol without the formation of difficult-to-remove impurities.
Large-scale synthesis considerations have also led to the development of continuous flow processes that utilize novel reactor designs optimized for solriamfetol production [1]. These systems employ specialized heat exchangers and mixing technologies that ensure uniform reaction conditions throughout the process stream, resulting in improved product quality and reduced batch-to-batch variability. The continuous flow approach also enables better control of reaction parameters such as residence time and temperature profiles, leading to enhanced process robustness.
[image:1]
Catalytic and Reaction Condition Optimization
The optimization of catalytic systems and reaction conditions represents a critical aspect of solriamfetol manufacturing, directly impacting product yield, purity, and overall process economics [1] [8]. Extensive research has focused on identifying optimal catalytic systems that promote the desired transformations while minimizing the formation of process-related impurities and degradation products.
Role of Tin Catalysts in Carbamate Formation
While the primary carbamate formation reaction in solriamfetol synthesis typically employs sodium cyanate under acidic conditions, recent investigations have explored the potential role of tin-based catalysts in enhancing reaction efficiency and selectivity [1] [8]. Although tin catalysts are not extensively detailed in the current solriamfetol literature, their known reactivity in carbamate formation reactions suggests potential applications in process optimization.
Tin catalysts, particularly tin(II) and tin(IV) compounds, have demonstrated significant activity in promoting carbamate formation reactions through their ability to activate both nucleophilic and electrophilic components [8]. In the context of solriamfetol synthesis, tin catalysts could potentially facilitate the reaction between D-phenylalaninol and cyanate species by coordinating to the alcohol oxygen atom, thereby increasing its nucleophilicity toward the electrophilic carbon center of the cyanate.
-
Solriamfetol | CAS 178429-62-4 | LGC Standards
Buy Solriamfetol, CAS number: 178429-62-4, online for pharmaceutical analytical testing. The highest quality reference standards for reliable results.
-
Buy Solriamfetol | 178429-62-4 | >98% – smolecule.com
Description Solriamfetol is a pharmacological compound primarily used as a …
-
Solriamfetol | CAS No. 178429-62-4 | Clearsynth
Buy Solriamfetol CAS No- 178429-62-4, a high quality product from Clearsynth, a Leading Manufacturer and Exporter of Solriamfetol accompanied by Certificate of Analysis. Click to check details.
-
CAS No : 178429-62-4 | Product Name : Solriamfetol
Solriamfetol is a selective dopamine and norepinephrine reuptake inhibitor used …
-
Reviews: 5
-
Catalogue number: PA 19 1750000
-
CAS Number: 178429-62-4
-
Chemical name: Solriamfetol
-
-
Buy Solriamfetol (EVT-284158) | 178429-62-4 – evitachem.com
Solriamfetol is a novel psychostimulant drug that has been recently approved by the US Food and Drug Administration (FDA) for the treatment of excessive daytime sleepiness (EDS) associated with …
-
Buy Solriamfetol | CAS 178429-62-4 | QCS Standards
HOME PRODUCTS SERVICES WHY QCS FAQ NEWS & BLOGS CONTACT US LOG IN REGISTER CART (0) 中文版 Espanha Portugal PRODUCT DETAIL PRODUCT DETAIL All Products > …
-
Solriamfetol |CAS 178429-62-4|DC Chemicals
Solriamfetol (JZP-110) is a selective dopamine and norepinephrine reuptake inhibitor with robust wake-promoting effects. Solriamfetol binds to dopamine and norepinephrine transporters, so it can inhibit …
-
CAS 178429-62-4 Solriamfetol – Globs Chemical LTD
Home Products Solriamfetol 🧪 Phenylethylamines Solriamfetol CAS: 178429-62-4 In Stock – Ready to Ship ⚡High Purity: 98% 🚚Lead Time: days
-
Solriamfetol | CAS NO.:178429-62-4 | GlpBio
Solriamfetol (JZP-110) is an orally active and selective dopamine and norepinephrine reuptake inhibitor with IC50s of 2.9 μM and 4.4 μM for dopamine and norepinephrine transporters, respectively.
Solriamfetol HCl | wake-promoting agent | CAS# 178429-62-4
Solriamfetol HCl (YKP-10A; ADXN-05; R228060; SKL-N-05; JZP-11; Sunosi), the hydrochloride salt of Solriamfetol which is a wake-promoting agent, is a norepinephrine–dopamine inhibitor that was



Reviews
There are no reviews yet.