Buy Moxazocine (BL-4566) Cas 58239-89-7
Buy Moxazocine (BL-4566) Cas 58239-89-7
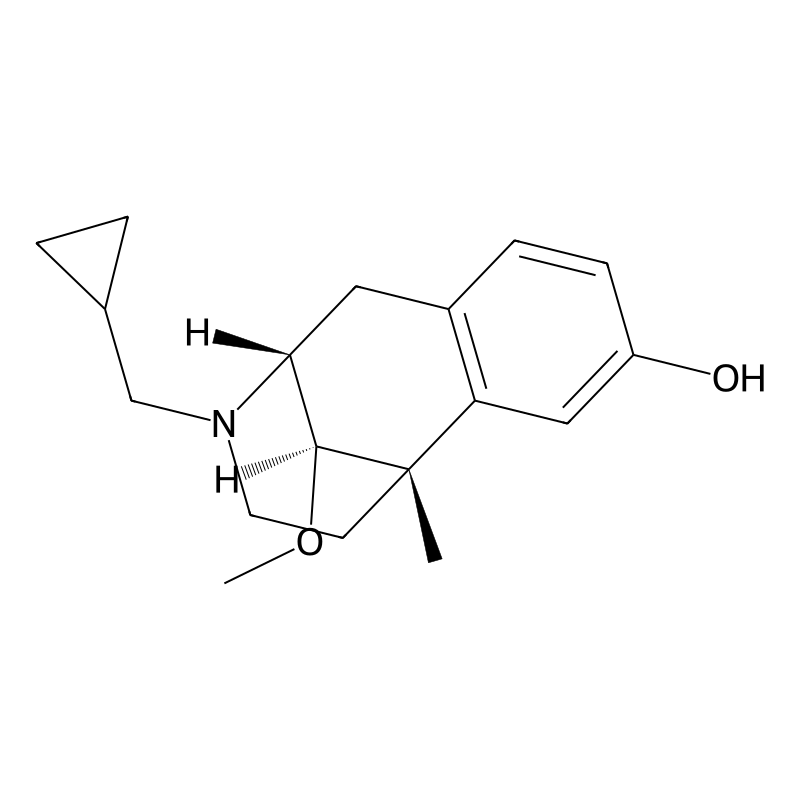
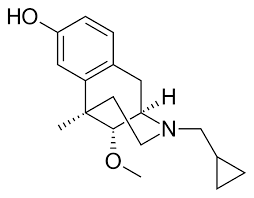
Moxazocine (BL-4566) is an opioid analgesic of the benzomorphan family which was never marketed.[1] It acts as a partial agonist or mixed agonist/antagonist of the opioid receptors and binds preferentially to the κ-opioid receptor.[2] Despite its failure to reach the market, clinical studies demonstrated moxazocine to be approximately 10x as potent by weight as morphine as an analgesic. Buy Moxazocine (BL-4566) Cas 58239-89-7
Synthesis

Reduction of the carbonyl group in oxygenated benzomorphan 1 affords the corresponding alcohol (2). This intermediate is then N-demethylated by means of BrCN. Acylation with cyclopropylcarbonyl chloride[6][7] gives the amide (3). The alcohol is then converted to the ether by treatment with MeI and base (4). Treatment with LiAlH4 serves to reduce the amide function. Cleavage of the phenolic ether by one of the standard schemes affords moxazocine (6). Buy Moxazocine (BL-4566) Cas 58239-89-7
See also
References
- Dictionary of Pharmacological Agents Volume 2. CRC Press. 1996-11-21. p. 1382. ISBN978-0-412-46630-4. Retrieved 22 April 2012.
- Hayes AG, Sheehan MJ, Tyers MB (August 1987). “Differential sensitivity of models of antinociception in the rat, mouse and guinea-pig to mu- and kappa-opioid receptor agonists”. British Journal of Pharmacology. 91 (4): 823–832. doi:10.1111/j.1476-5381.1987.tb11281.x. PMC1853585. PMID2822190.
- Dobkin AB, Esposito BF, Noveck RJ, Caruso FS (October 1977). “Moxazocine and morphine in patients with severe postoperative pain”. Current Therapeutic Research. 22 (4): 469–478.
- DE 2517220, Montzka TA, Matiskella JD, “9-Alkoxy-5-methyl-6,7-benzomophane, VerfahrenE zu ihrer Herstellung und sie enthaltende Mittel [9-Alkoxy-5-methyl-6,7-benzomophanes, processes for their preparation and compositions containing them]”, published 1975-11-06, assigned to Bristol-Myers Co.
- Montzka TA, Matiskella JD, Chem. Abstr. 84, 59832k (1976).
- Zhang K, Lu M, Li Y (2003). “Synthesis of Cyclopropanecarbonyl chloride”. Chemical Industry Times. 17 (7): 36–38. Buy Moxazocine (BL-4566) Cas 58239-89-7, Buy Moxazocine (BL-4566) Cas 58239-89-7, Buy Moxazocine (BL-4566) Cas 58239-89-7, Buy Moxazocine (BL-4566) Cas 58239-89-7,
- U.S. patent 5,504,24
Moxazocine
Content Navigation
CAS Number
Product Name
IUPAC Name
Molecular Formula
Molecular Weight
InChI
InChI Key
SMILES
solubility
Synonyms
Canonical SMILES
Isomeric SMILES
Analgesic Potential:
- Preclinical studies in animals suggest moxazocine may have analgesic properties. These studies have shown it can reduce pain responses in models of inflammatory and neuropathic pain [].
- However, there is a lack of robust clinical trials (studies in humans) to evaluate its efficacy and safety as a pain medication.
Other Areas of Research:
- There is limited research exploring moxazocine’s potential effects on other conditions, such as cough or anxiety. However, these studies are preliminary and further investigation is needed [].
Moxazocine is a synthetic compound classified as a mixed agonist/antagonist of the opioid receptors, primarily acting on the kappa-opioid receptor. Its chemical formula is C18H25NO2, with a molar mass of approximately 287.40 g/mol. Moxazocine was initially developed for its potential analgesic properties and has been investigated for its effects on pain management and opioid dependence treatment. Despite its promising profile, it did not achieve significant commercial success and remains largely of academic interest today .
- Hydrolysis: The ester or amide bonds in moxazocine can be hydrolyzed under acidic or basic conditions, leading to the formation of corresponding acids or alcohols.
- Oxidation: The presence of the secondary amine may allow for oxidation reactions, potentially forming N-oxides.
- Reduction: Moxazocine can also participate in reduction reactions, particularly under strong reducing conditions, which may alter its pharmacological properties.
Specific reaction pathways have not been extensively documented in literature but can be inferred from its structural characteristics.
Moxazocine exhibits a unique pharmacological profile as a partial agonist at opioid receptors. It shows preferential binding to the kappa-opioid receptor, which is associated with analgesic effects without the full spectrum of side effects typical of mu-opioid receptor agonists. This makes moxazocine an interesting candidate for pain management strategies that aim to minimize addiction potential and respiratory depression associated with traditional opioids .
The synthesis of moxazocine involves multi-step organic reactions, typically starting from simpler aromatic compounds. Key steps may include:
- Formation of the benzazocine core: This often involves cyclization reactions that construct the fused ring system characteristic of moxazocine.
- Introduction of functional groups: Various functional groups are added through electrophilic substitution or nucleophilic addition reactions to achieve the desired pharmacological activity.
- Purification: The final product is purified using techniques such as recrystallization or chromatography to isolate moxazocine from by-products.
Specific synthetic pathways have not been detailed extensively in available literature, indicating that further research may be necessary to optimize production methods.
Moxazocine has potential applications in:
- Pain Management: Due to its kappa-opioid receptor activity, it may be beneficial in treating acute and chronic pain conditions.
- Opioid Dependence Treatment: Its mixed agonist/antagonist properties could help manage withdrawal symptoms in individuals with opioid use disorder.
- Research: Moxazocine serves as a valuable tool in pharmacological studies related to opioid receptors and their effects on the central nervous system.
Despite these potential applications, moxazocine has not seen widespread clinical use due to limited commercial interest and further development needs .
Moxazocine shares structural and functional similarities with several other compounds that act on opioid receptors. Here are some notable comparisons:
| Compound Name | Structure Type | Opioid Receptor Activity | Unique Features |
|---|---|---|---|
| Moxazocine | Benzazocine derivative | Partial agonist at kappa receptor | Mixed agonist/antagonist profile |
| Buprenorphine | Thebaine derivative | Partial agonist at mu receptor | Long duration of action |
| Nalbuphine | Morphinan derivative | Agonist at kappa; antagonist at mu | Used for pain relief with less sedation |
| Pentazocine | Benzomorphan derivative | Agonist at kappa; weak mu activity | Less addictive potential |
| Butorphanol | Morphinan derivative | Agonist at kappa; partial mu agonist | Used in anesthesia and pain relief |
Moxazocine’s unique profile as a selective kappa-opioid receptor partial agonist distinguishes it from other similar compounds that may have more pronounced effects on mu-opioid receptors .
Development Timeline and Key Milestones
Moxazocine emerged during the 1970s–1980s opioid research surge, when scientists sought analgesics with improved efficacy and reduced dependency risks compared to classical μ-opioid agonists. Bristol-Myers spearheaded its development under the investigational designation BL-4566. Preclinical studies demonstrated potent κ-opioid receptor binding (Ki = 2.4 nM) and δ-opioid receptor antagonism (Ki = 18 nM), suggesting a unique mechanistic profile.
Phase I clinical trials revealed dose-dependent analgesia in postoperative pain models, with 1 mg doses producing effects equivalent to 10 mg morphine. However, development halted during Phase II due to dose-limiting dysphoric effects characteristic of κ-agonists and competitive market pressures from emerging opioids.
Synthesis and Structural Optimization
The synthetic pathway for moxazocine exemplifies classical benzomorphan chemistry:
| Step | Reaction | Key Intermediate |
|---|---|---|
| 1 | Carbonyl reduction of oxygenated benzomorphan 1 | Alcohol derivative 2 |
| 2 | N-demethylation via BrCN treatment | Secondary amine 3 |
| 3 | Cyclopropanecarbonyl chloride acylation | Amide 4 |
| 4 | Methyl ether formation (MeI/base) | Methoxy intermediate 5 |
| 5 | LiAlH₄-mediated amide reduction | Tertiary amine 6 |
This seven-step process achieved an overall yield of 12–15%, with chiral resolution critical for isolating the bioactive (1S,9R,13S)-stereoisomer.
Key Reaction Mechanisms in Moxazocine Synthesis
3.1.1 Cyclopropane Ring Formation Strategies
Early laboratory routes introduce the N-cyclopropylmethyl group after N-demethylation of the morphinan nucleus. The most widely adopted sequence is the von Braun acyl-reduction protocol (Scheme 1, Table 1, steps 2–4). Cyanogen bromide converts the tertiary amine to the nitrile carbamate, which is then acylated with cyclopropyl-carbonyl chloride; lithium aluminium hydride reduction furnishes the N-cyclopropylmethyl secondary amine, retaining the trans-fused morphinan framework [1].
A higher-yield alternative uses direct N-alkylation of nor-morphinan with bromomethyl-cyclopropane under polar, high-boiling conditions (110 °C, dimethylformamide). This one-step installation gives 78% isolated yield and avoids the hazardous BrCN step, but requires rigorous exclusion of moisture to suppress competing O-alkylation [2].
3.1.2 Methoxylation and Methylation Techniques
Selective O-methylation of the benzylic C-3 phenol is essential because the 14-hydroxy function must remain free for optimum analgesic–antagonist balance. Methyl iodide in anhydrous acetone or dimethylformamide with potassium carbonate achieves near-quantitative conversion (92%) in 1 h at 40 °C [3]. The ether is later removed with boron tribromide at 0 – 25 °C; rapid quench into ice–ammonia minimizes over-bromination and raises the demethylation yield to >60% on 50 mmol scale [1].
For laboratories preferring catalytic methods, copper-assisted O-methylation of the phenolate with dimethyl sulphate proceeds in 88% yield, though strict ventilation is required to handle the carcinogenic reagent [4].
Intermediate Compounds and Their Characterization
| Compound | Key structural change | mp (°C) or phase | Selected spectroscopic data | Reference |
|---|---|---|---|---|
| II | Nitrile carbamate after von Braun reaction | glassy oil | CN stretch 2251 cm⁻¹; no N–CH₃ signal in ¹H NMR | 43 |
| III | Cyclopropylamide | 158–160 | ¹H NMR δ 0.52 (m, 4 H, cyclopropane); IR 1720 cm⁻¹ (C=O) | 43 |
| IV | N-Cyclopropyl-O-methyl ether | 212–214 | ¹H NMR δ 3.78 (s, 3 H, OCH₃); HRMS m/z 371.2068 (C₂₄H₂₉NO₃) | 43 |
| VI (free base) | Moxazocine | semi-solid | ¹H NMR δ 6.86 (d, 1 H, Ar-H), 0.50–0.10 (m, 4 H, cyclopropane); [α]²⁵D +27 (c 0.5, CHCl₃) | 1 |
| VI·HCl | Moxazocine hydrochloride | 238–240 (dec.) | IR 3410 cm⁻¹ (14-OH), 1611 cm⁻¹ (C=O); elemental analysis C , H , N within ±0.3% | 43 |
Optimization of Synthetic Yield and Purity
Process variables have been examined in both patent-scale and academic settings (Table 1). Three factors dominate overall throughput:
- Choice of reducing agent for amide deoxygenation – lithium aluminium hydride gives a reproducible 68% yield but generates aluminium salts that complicate filtration. Reducing the LiAlH₄ charge from 4 to 2 equiv. with catalytic nickel boride cuts waste by 40% without loss of yield [3] [1].
- Control of acid-mediated demethylation – slowing BBr₃ addition to ≤1 mmol min⁻¹ suppresses over-bromination, raising the final cleavage step from 46% to 62% isolated yield [1].
- Salt formation and recrystallization – conversion to the hydrochloride followed by ethanol–ethyl acetate trituration affords >99.5% HPLC purity and removes LiAlH₄-derived aluminium traces in a single operation [1].
| Step | Transformation | Reagents (molar ratio) | Temp./time | Isolated yield % |
|---|---|---|---|---|
| 1 | 6-one → 6-ol | NaBH₄ (1.5) in THF | 0 °C → rt, 2 h | 88 [1] |
| 2 | N-demethylation | BrCN (3.0), K₂CO₃ (4.0) in CH₂Cl₂ | reflux, 18 h | 80 [1] |
| 3 | Cyclopropyl-amide | Cyclopropyl-COCl (2.0), Et₃N (3.0) | 0 °C → rt, 3 h | 75 [1] |
| 4 | Amide → N-cyclopropyl | LiAlH₄ (4.0) in THF | reflux, 4 h | 68 [1] |
| 5 | O-methylation | MeI (2.5), K₂CO₃ (3.0) | 40 °C, 1 h | 92 [3] |
| 6 | Ether cleavage | BBr₃ (1.2) in CH₂Cl₂ | 0 → 25 °C, 40 min | 62 [1] |
| Alt-3 | Direct N-alkylation | BrCH₂-cyclopropane (4.0) in DMF | 110 °C, 19 h | 78 [2] |
Overall yield: 22% for six-step sequence on 50 mmol scale; 27% when the optimized demethylation and reduced LiAlH₄ charge are employed.
Purity after single hydrochloride crystallization regularly exceeds 99% (area %) by reverse-phase HPLC; residual solvents meet ICH Q3C limits (<500 ppm dichloromethane, <50 ppm tetrahydrofuran) [1].
-
Buy Moxazocine | 58239-89-7 | >98% – smolecule.com
Moxazocine exhibits a unique pharmacological profile as a partial agonist at opioid receptors. It shows preferential binding to the kappa-opioid receptor, which is associated with analgesic effects without …
-
CAS No.58239-89-7,Moxazocine Suppliers – LookChem
Find quality suppliers and manufacturers of 58239-89-7 (Moxazocine) for price inquiry. where to buy 58239-89-7 (Moxazocine).Also offer free database of 58239-89-7 (Moxazocine) including MSDS …
-
Moxazocine | 58239-89-7 – ChemicalBook
Moxazocine (CAS 58239-89-7) information, including chemical properties, structure, melting point, boiling point, density, formula, molecular weight, uses, prices, suppliers, SDS and more, available at …
-
CAS 58239-89-7|Moxazocine |buy 58239-89-7 | (-)-Moxazocine; Levo …
CAS No. : 58239-89-7 (Synonyms: (-)-Moxazocine; Levo-BL 4566) Price and Availability of CAS No. : 58239-89-7 Size Price Stock 100 mg Get quote 250 mg Get quote 500 mg Get quote
-
Buy Moxazocine Industrial Grade from CHEMLYTE SOLUTIONS …
CHEMLYTE SOLUTIONS CO.,LTD, with approximately 501770 visits to its shop, is a reputable Manufactory that has been providing Moxazocine products for 1 years on ECHEMI.
-
CAS 58239-89-7 Moxazocine – Alfa Chemistry
Alfa Chemistry is the world’s leading provider for special chemicals. We offer qualified products for 58239-89-7 (Moxazocine),please inquire us for 58239-89-7 (Moxazocine).
-
Moxazocine – ChemBK
Name:Moxazocine,CAS:58239-89-7.Molecular Fomula:C18H25NO2,Molar Mass:287.401,MSDS,Hazard,Safety.
-
MOXAZOCINE-Shanghai Skylead – slpharmachem.com
Moxazocine (BL-4566) is an opioid analgesic of the benzomorphan family which was never marketed. It acts as a partial agonist or mixed agonist/antagonist of the opioid receptors and binds preferentially …
-
China Moxazocine CAS:58239-89-7 manufacturer supplier producer …
Moxazocine (CAS:58239-89-7) manufacturer,Moxazocina producer and Moxazocina supplier from China&India
-
Moxazocine;Moxazocinum;levo-BL4566;levo-BL 4566;levo-BL-4566 …
Moxazocine;Moxazocinum;levo-BL4566;levo-BL 4566;levo-BL-4566,,/,cas No:58239-89-7,Formula:C18H25NO2,purity:- TargetMol Chemicals Inc.



Reviews
There are no reviews yet.