Buy Phenomorphan Cas 468-07-5
Phenomorphan is an opioid analgesic. It is not currently used in medicine, but has similar side-effects to other opiates, which include itching, nausea and respiratory depression.
Phenomorphan is a highly potent drug due to the N-phenethyl group, which boosts affinity to the μ-opioid receptor, and so phenomorphan is around 10x more potent than levorphanol, which is itself 6-8x the potency of morphine. Other analogues where the N-(2-phenylethyl) group has been replaced by other aromatic rings[3] are even more potent, with the N-(2-(2-furyl)ethyl) and the N-(2-(2-thienyl)ethyl) analogues being 60x and 45x stronger than levorphanol, respectively. Buy Phenomorphan Cas 468-07-5, Buy Phenomorphan Cas 468-07-5, Buy Phenomorphan Cas 468-07-5, Buy Phenomorphan Cas 468-07-5, Buy Phenomorphan Cas 468-07-5, Buy Phenomorphan Cas 468-07-5, Buy Phenomorphan Cas 468-07-5, Buy Phenomorphan Cas 468-07-5, Buy Phenomorphan Cas 468-07-5, Buy Phenomorphan Cas 468-07-5, Buy Phenomorphan Cas 468-07-5, Buy Phenomorphan Cas 468-07-5, Buy Phenomorphan Cas 468-07-5, Buy Phenomorphan Cas 468-07-5, Buy Phenomorphan Cas 468-07-5
See also
- 14-Cinnamoyloxycodeinone
- 14-Phenylpropoxymetopon
- 7-PET
- N-Phenethylnormorphine
- N-Phenethylnordesomorphine
- N-Phenethyl-14-ethoxymetopon
- RAM-378
- Ro4-1539
References
- Anvisa (2023-03-31). “RDC Nº 784 – Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial” [Collegiate Board Resolution No. 784 – Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-16.
- US patent 2885401, Grussner A, Hellerbach J, Schnider O, “Process for making morphinan derivatives and products available thereby”, published 1956-03-22, issued 1959-05-05
- US patent 2970147, Andre Grussner, Joseph Hellerbach, Otto Schnider, “3-hydroxy-N-(heterocyclic-ethyl)-morphinans”, published 1958-11-26, issued 1961-01-31
- Hellerbach J, Schnider O, Besendorf H, Pellmont B (1966). Synthetic Analgesics: Part IIA. Morphinans. International series of monographs on organic chemistry. Pergamon Press. Buy Phenomorphan Cas 468-07-5
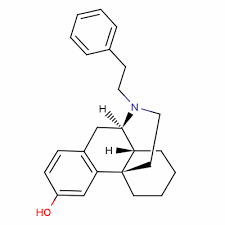
Phenomorphan
- Chemical Name:Phenomorphan
- CAS No.:468-07-5
- Molecular Formula:C24H29NO
- Molecular Weight:347.5
- Hs Code.:
- European Community (EC) Number:207-403-2
- UNII:26HXE4B73P
- DSSTox Substance ID:DTXSID70878657
- Wikipedia:Phenomorphan
- Wikidata:Q7181393
- NCI Thesaurus Code:C166547
- ChEMBL ID:CHEMBL2105429
- Mol file:468-07-5.mol
Synonyms:Phenomorphan;NIH 7274;468-07-5;Fenomorfano;Phenomorphane;Phenomorphanum;Fenomorfano [INN-Spanish];Phenomorphane [INN-French];Phenomorphanum [INN-Latin];3-HYDROXY-N-PHENETHYLMORPHINAN;Phenomorphan [INN:BAN:DCF];MCL 112;Morphinan, 3-hydroxy-N-phenethyl-;EINECS 207-403-2;UNII-26HXE4B73P;26HXE4B73P;IDS-NP-009;DEA No. 9647;Morphinan-3-ol, 17-phenethyl-, (-)-;2H-10,4a-Iminoethanophenanthren-6-ol, 1,3,4,9,10,10a,hexahydro-11-phenethyl-;17-(2-Phenylethyl)morphinan-3-ol;Morphinan-3-ol, 17-(2-phenylethyl)-;PHENOMORPHAN [MI];PHENOMORPHAN [INN];SCHEMBL79352;Phenomorphan [BAN:DCF:INN];C24H29NO;CHEMBL2105429;DTXSID70878657;3-hydroxy-N-(2-phenylethyl)morphinan;LS-92054;Q7181393;2H-10,4A-IMINOETHANOPHENANTHREN-6-OL, 1,3,4,9,10,10A-HEXAHYDRO-11-PHENETHYL-
 This product is a nationally controlled contraband, and the Lookchem platform doesn’t provide relevant sales information.
This product is a nationally controlled contraband, and the Lookchem platform doesn’t provide relevant sales information.
- Vapor Pressure:3.13E-11mmHg at 25°C
- Melting Point:243-245°
- Boiling Point:515.1°Cat760mmHg
- PKA:pKa 7.30(50% aq EtOH) (Uncertain)
- Flash Point:260.6°C
- PSA:23.47000
- Density:1.18g/cm3
- LogP:4.63120
- XLogP3:5.1
- Hydrogen Bond Donor Count:1
- Hydrogen Bond Acceptor Count:2
- Rotatable Bond Count:3
- Exact Mass:347.224914549
- Heavy Atom Count:26
- Complexity:487
- Purity/Quality:
- Pictogram(s):
- Hazard Codes:
- MSDS Files:
- SDS file from LookChem
- Canonical SMILES:C1CCC23CCN(C(C2C1)CC4=C3C=C(C=C4)O)CCC5=CC=CC=C5
- Isomeric SMILES:C1CC[C@@]23CCN([C@@H]([C@@H]2C1)CC4=C3C=C(C=C4)O)CCC5=CC=CC=C5
- UsesControlled substance (opiate).
There total 9 articles about Phenomorphan which guide to synthetic route it. The literature collected by LookChem mainly comes from the sharing of users and the free literature resources found by Internet computing technology. We keep the original model of the professional version of literature to make it easier and faster for users to retrieve and use. At the same time, we analyze and calculate the most feasible synthesis route with the highest yield for your reference as below:
- Guidance literature:
-
With lithium aluminium tetrahydride; In tetrahydrofuran; at 20 ℃;
Reference yield:
- Guidance literature:
-
Multi-step reaction with 2 steps1: Et3N / CH2Cl2 / 0 – 20 °C2: LiAlH4 / tetrahydrofuran / 20 °CWith lithium aluminium tetrahydride; triethylamine; In tetrahydrofuran; dichloromethane;
Phenomorphan is a synthetic compound belonging to the class of morphinans, which are derivatives of morphine. It is characterized by its complex molecular structure and pharmacological properties, making it a subject of interest in medicinal chemistry and pharmacology. Phenomorphan acts primarily as an opioid agonist, influencing the central nervous system and modulating pain perception.
Phenomorphan can be synthesized from various precursors, including naturally occurring opiates. Its synthesis typically involves multiple steps of chemical transformations, allowing for the introduction of specific functional groups that enhance its pharmacological profile.
Phenomorphan is classified as an opioid analgesic. It is structurally related to other morphinans and has been studied for its potential therapeutic applications in pain management and anesthesia.
Methods
The synthesis of phenomorphan involves several key steps, often starting from commercially available materials. Common methods include:
- Alkylation: Alkylation reactions are used to introduce substituents onto the nitrogen atom of the morphinan skeleton.
- Cyclization: This step often involves forming rings through intramolecular reactions, which are crucial for establishing the compound’s three-dimensional structure.
- Reduction: Various reduction techniques are employed to convert double bonds into single bonds or to modify functional groups.
Technical Details
For instance, one method described in literature involves synthesizing ortho-c oxide-bridged phenylmorphans through alkylation with allyl bromide followed by cyclization using formic and phosphoric acids . This method yields intermediates that can be further modified to produce phenomorphan derivatives.
Structure
Phenomorphan possesses a complex molecular structure characterized by a phenyl ring and a morphinan backbone. The specific arrangement of atoms and functional groups defines its biological activity.
Data
The molecular formula for phenomorphan is C18H23NO, indicating it contains 18 carbon atoms, 23 hydrogen atoms, one nitrogen atom, and one oxygen atom. The molecular weight is approximately 273.39 g/mol.
Reactions
Phenomorphan undergoes various chemical reactions that are essential for its synthesis and modification:
- N-Methylation: This reaction introduces a methyl group onto the nitrogen atom, enhancing the compound’s potency.
- Bromination: Selective bromination at specific carbon positions allows for further functionalization.
- Hydrogenation: This process reduces double bonds within the molecule, impacting its pharmacological properties.
Technical Details
For example, N-bromoacetamide can be used to brominate phenomorphan derivatives at specific positions, followed by reduction with sodium cyanoborohydride to yield saturated compounds .
Process
Phenomorphan primarily acts as an agonist at mu-opioid receptors in the brain and spinal cord. Upon binding to these receptors, it mimics the effects of endogenous opioids such as endorphins.
Data
The activation of mu-opioid receptors results in decreased neurotransmitter release, leading to reduced pain perception and increased feelings of euphoria. This mechanism underlies its potential use in pain management therapies.
Physical Properties
Phenomorphan is typically a white crystalline solid with moderate solubility in organic solvents. Its melting point and boiling point can vary based on purity and specific structural modifications.
Chemical Properties
- Solubility: Soluble in ethanol and methanol; limited solubility in water.
- Stability: Generally stable under standard laboratory conditions but may degrade under extreme pH or temperature conditions.
- Reactivity: Reacts with strong acids or bases during synthesis or degradation processes.
Relevant analyses indicate that modifications to its structure can significantly affect these properties, influencing both its therapeutic efficacy and safety profile.
Scientific Uses
Phenomorphan has potential applications in several fields:
- Pain Management: Due to its opioid activity, it may be used in clinical settings for treating acute or chronic pain.
- Anesthesia: Its properties could make it suitable for use in anesthetic protocols.
- Research: Phenomorphan serves as a valuable tool in pharmacological research aimed at understanding opioid receptor interactions and developing new analgesics.
Properties
CAS Number
Product Name
IUPAC Name
Molecular Formula
Molecular Weight
InChI
InChI Key
Canonical SMILES
Isomeric SMILES
-
Buy Online CAS Number 468-07-5 – TRC – Phenomorphan (1.0 …
Purchase online CAS Number 468-07-5 – TRC – Phenomorphan (1.0 mg/mL in Methanol). High Quality CRMs, Reference Materials, Proficiency Testing & More at LGC Standards.
-
Buy Phenomorphan |Cas 468-07-5 –
Phenomorphan is a synthetic compound belonging to the class of morphinans, which are derivatives of morphine. It is characterized by its complex molecular structure and pharmacological properties, …
-
phenomorphan CAS#: 468-07-5 – ChemicalBook
Request For Quotation phenomorphan Suppliers list … Tag:phenomorphan (468-07-5) Related Product Information phenomorphan N-methylmorphinan phenazocine levorphanol levophenacylmorphan
-
Phenomorphan | CAS# 468-07-5 | opioid analgesic | MedKoo
Phenomorphan is an opioid analgesic. It is not currently used in medicine, but has similar side-effects to other opiates, which include itching, nausea and respiratory depression.
-
468-07-5 (Phenomorphan),D12688,MCL 112 | Kuujia.com
Find 468-07-5 (Phenomorphan) chemical properties, uses, structure, security information, melting point, boiling point, density, molecular formula, prices, suppliers, and latest news & literatures on Kuujia.com.
-
CAS: 468-07-5 – phenomorphan | CymitQuimica
Phenomorphan (1.0 mg/mL in Methanol) Controlled Product CAS:468-07-5 Formula: C 24 H 29 NO Color and Shape: Single Solution Molecular weight: 347.49
-
468-07-5 phenomorphan – ChemNet
Chemical name:phenomorphan ; CAS NO:468-07-5; Molecular Formula:C<sub>24</sub>H<sub>29</sub>NO; Molecular Weight:347.4932; EINECS:207-403-2
-
Phenomorphan|468-07-5|lookchem
We keep the original model of the professional version of literature to make it easier and faster for users to retrieve and use. At the same time, we analyze and calculate the most feasible synthesis route …
-
CAS # 468-07-5, Phenomorphan: more information.
chemBlink provides more information about CAS # 468-07-5, Phenomorphan (molecular formula: C24H29NO).
-




Reviews
There are no reviews yet.