Buy Seproxetine (S)-norfluoxetine Cas 126924-38-7
Buy Seproxetine (S)-norfluoxetine Cas 126924-38-7
Seproxetine, also known as (S)-norfluoxetine, is a selective serotonin reuptake inhibitor (SSRI).[1][2] It is the S enantiomer of norfluoxetine, the main active metabolite of the widely used antidepressant fluoxetine;[3] it is nearly 4 times more selective for stimulating neurosteroid synthesis relative to serotonin reuptake inhibition than fluoxetine.[4] It is formed through the demethylation, or removal of a methyl group, of fluoxetine.[5] Seproxetine is both an inhibitor of serotonin and dopamine transporters, 5-HT2A and 5-HT2C receptors.[6] It was being investigated by Eli Lilly and Company as an antidepressant; however, it inhibited the KvLQT1 protein, which is responsible for the management of the QT interval. This is the time it takes for the heart to contract and recover. Due to the inhibition, the QT interval was prolonged, which could lead to significant cardiac side complications.[7] Due to this, development of the medication was discontinued.[1] Tests on its efficacy found that it was equivalent to fluoxetine, but sixteen times more powerful than the R enantiomer of norfluoxetine. Buy Seproxetine (S)-norfluoxetine Cas 126924-38-7
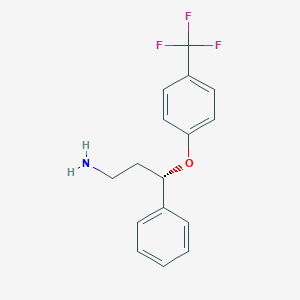
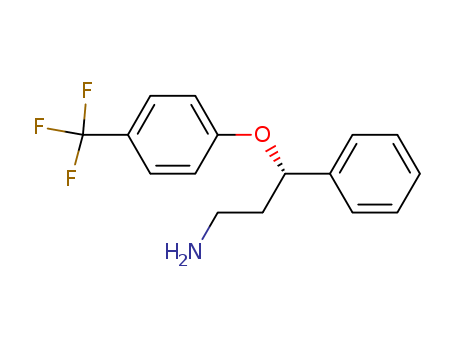
S)-3-PHENYL-3-(4-TRIFLUOROMETHYL-PHENOXY)-PROPYLAMINE
Content Navigation
CAS Number
Product Name
IUPAC Name
Molecular Formula
Molecular Weight
InChI
InChI Key
SMILES
Synonyms
Canonical SMILES
Isomeric SMILES
Potential as an Antidepressant:
Seproxetine, also known as S-norfluoxetine, is a selective serotonin reuptake inhibitor (SSRI). Although its development as an antidepressant was discontinued due to safety concerns, research suggests it possesses qualities that could be relevant to future antidepressant development.
- Potency: Studies have shown that seproxetine is more potent than fluoxetine, the antidepressant from which it is derived, and significantly more potent than the other enantiomer of norfluoxetine. [1]
- Mechanism of Action: Beyond inhibiting serotonin reuptake, seproxetine also interacts with dopamine transporters and 5-HT2A/2C receptors, potentially offering a broader range of action compared to other SSRIs. [1]
Exploring Improved Efficacy:
- Charge-Transfer Complexes: Studies are investigating the use of charge-transfer complexes to enhance seproxetine’s effectiveness while potentially mitigating its side effects. [2] Buy Seproxetine (S)-norfluoxetine Cas 126924-38-7, Buy Seproxetine (S)-norfluoxetine Cas 126924-38-7, Buy Seproxetine (S)-norfluoxetine Cas 126924-38-7, Buy Seproxetine (S)-norfluoxetine Cas 126924-38-7, Buy Seproxetine (S)-norfluoxetine Cas 126924-38-7, Buy Seproxetine (S)-norfluoxetine Cas 126924-38-7, Buy Seproxetine (S)-norfluoxetine Cas 126924-38-7, Buy Seproxetine (S)-norfluoxetine Cas 126924-38-7, Buy Seproxetine (S)-norfluoxetine Cas 126924-38-7, Buy Seproxetine (S)-norfluoxetine Cas 126924-38-7, Buy Seproxetine (S)-norfluoxetine Cas 126924-38-7, Buy Seproxetine (S)-norfluoxetine Cas 126924-38-7, Buy Seproxetine (S)-norfluoxetine Cas 126924-38-7, Buy Seproxetine (S)-norfluoxetine Cas 126924-38-7, Buy Seproxetine (S)-norfluoxetine Cas 126924-38-7, Buy Seproxetine (S)-norfluoxetine Cas 126924-38-7
Importance of Continued Research:
While seproxetine is not currently used as a medication, it serves as a valuable research tool for understanding the mechanisms of action of antidepressants and exploring potential avenues for developing more effective and safer treatments for depression.
Citations:
- Increasing the Efficacy of Seproxetine as an Antidepressant Using Charge–Transfer Complexes:
- Increasing the Efficacy of Seproxetine as an Antidepressant Using Charge-Transfer Complexes:
(S)-3-Phenyl-3-(4-trifluoromethyl-phenoxy)-propylamine, commonly known as (S)-fluoxetine, is a chiral compound with significant pharmacological properties. It is classified as an antidepressant and is primarily recognized for its role as a selective serotonin reuptake inhibitor (SSRI). The chemical formula is C₁₇H₁₈F₃NO, and it has a molecular weight of approximately 309.33 g/mol. This compound features a trifluoromethyl group attached to a phenoxy moiety, contributing to its unique properties and biological activity .
- N-Methylation: The amine can be methylated to form N-methyl derivatives, which are crucial for enhancing its pharmacological activity.
- Acylation: The amine group can react with acyl chlorides to form amides, potentially modifying its biological activity.
- Oxidation: The phenolic part of the molecule can undergo oxidation reactions, impacting its stability and reactivity.
These reactions are essential for modifying the compound’s structure to enhance its therapeutic efficacy or reduce side effects .
(S)-3-Phenyl-3-(4-trifluoromethyl-phenoxy)-propylamine exhibits notable biological activity primarily as an antidepressant. Its mechanism involves the inhibition of serotonin reuptake in the synaptic cleft, thereby increasing serotonin levels in the brain. This action contributes to its effectiveness in treating major depressive disorder and other mood disorders . Additionally, it has been shown to have a high affinity for serotonin transporters, making it a potent SSRI .
The synthesis of (S)-3-Phenyl-3-(4-trifluoromethyl-phenoxy)-propylamine typically involves several key steps:
- Formation of the Phenoxy Group: The initial step often includes the synthesis of the 4-trifluoromethylphenol derivative.
- Amine Formation: The phenolic compound is then reacted with an appropriate alkylating agent to introduce the propylamine chain.
- Resolution of Enantiomers: Given that fluoxetine exists as a racemic mixture, methods such as chiral chromatography or enzymatic resolution are employed to isolate the (S) enantiomer .
These methods are crucial for producing high-purity compounds suitable for pharmaceutical applications.
(S)-3-Phenyl-3-(4-trifluoromethyl-phenoxy)-propylamine is primarily used in:
- Pharmaceuticals: As an antidepressant medication under various brand names, including Prozac.
- Research: Studied for its effects on serotonin pathways and potential applications in treating anxiety disorders and obsessive-compulsive disorder.
Its unique structure allows for ongoing research into additional therapeutic uses beyond depression .
Interaction studies have demonstrated that (S)-3-Phenyl-3-(4-trifluoromethyl-phenoxy)-propylamine can interact with various neurotransmitter systems beyond serotonin. Notably:
- Dopaminergic Systems: Some studies suggest potential interactions with dopamine receptors, which could influence mood regulation.
- Cytochrome P450 Enzymes: It has been shown to be a substrate and inhibitor of several cytochrome P450 enzymes, impacting drug metabolism and interactions .
These interactions highlight the need for careful consideration when co-administering this compound with other medications.
Several compounds share structural similarities with (S)-3-Phenyl-3-(4-trifluoromethyl-phenoxy)-propylamine. Here are some notable examples:
| Compound Name | Structure | Unique Features |
|---|---|---|
| (R)-Fluoxetine | C₁₇H₁₈F₃NO | Enantiomer of (S)-fluoxetine; less potent as an SSRI |
| Norfluoxetine | C₁₆H₁₈F₃NO | Active metabolite of fluoxetine; longer half-life |
| Paroxetine | C₁₉H₂₁F₂N | SSRI with different side effects; more potent at serotonin receptors |
| Sertraline | C₁₈H₂₄ClN | Another SSRI; distinct mechanism involving dopamine receptors |
The uniqueness of (S)-3-Phenyl-3-(4-trifluoromethyl-phenoxy)-propylamine lies in its specific trifluoromethyl substitution, which enhances its binding affinity to serotonin transporters compared to these similar compounds .
UNII 25CO3X0R31
Mechanism of Action
Other CAS 126924-38-7
Metabolism Metabolites
Wikipedia
Biological Half Life
Dates
Seproxetine
- Chemical Name:Seproxetine
- CAS No.:126924-38-7
- Molecular Formula:C16H16 F3 N O
- Molecular Weight:295.304
- Hs Code.:2922299090
- UNII:25CO3X0R31
- DSSTox Substance ID:DTXSID001019529,DTXSID50891581
- Nikkaji Number:J511.686D
- Wikipedia:Seproxetine
- Wikidata:Q63395592
- NCI Thesaurus Code:C75177
- Pharos Ligand ID:9V2X6ZD2JJ8N
- Metabolomics Workbench ID:149938
- ChEMBL ID:CHEMBL465123
- Mol file:126924-38-7.mol
Synonyms:(S)-Norfluoxetine;SEPROXETINE;126924-38-7;(3S)-3-phenyl-3-[4-(trifluoromethyl)phenoxy]propan-1-amine;Seproxetine [INN];(S)-3-PHENYL-3-(4-TRIFLUOROMETHYL-PHENOXY)-PROPYLAMINE;UNII-25CO3X0R31;Norfluoxetine, (s)-;(S)-3-phenyl-3-(4-(trifluoromethyl)phenoxy)propan-1-amine;CHEMBL465123;25CO3X0R31;S-Norfluoxetine;Benzenepropanamine, .gamma.-[4-(trifluoromethyl)phenoxy]-, (.gamma.S)-;BENZENEPROPANAMINE, .GAMMA.-(4-(TRIFLUOROMETHYL)PHENOXY)-, (.GAMMA.S)-;D08AWN;SEPROXETINE [WHO-DD];SCHEMBL272873;(S)-3-(4-Trifluoromethylphenoxy)-3-phenylpropylamine;DTXSID50891581;WIQRCHMSJFFONW-HNNXBMFYSA-N;DTXSID001019529;BDBM50254790;AKOS027384574;DB06731;NFL_296.1257_14.5;J-005451;Q63395592
- Business phase:
- The product has achieved commercial mass production*data from LookChem market partment
- Manufacturers and distributors:
-
- Manufacture/Brand
- Chemicals and raw materials
- Packaging
- price
- TRC
- (S)-Norfluoxetine
- 2.5mg
- $ 185.00
- SynQuest Laboratories
- 1-[(1S)-3-Amino-1-phenylpropoxy]-4-(trifluoromethyl)benzene 96%
- 250 mg
- $ 1193.00
- SynQuest Laboratories
- 1-[(1S)-3-Amino-1-phenylpropoxy]-4-(trifluoromethyl)benzene 96%
- 1 g
- $ 3195.00
- J&W Pharmlab
- (S)-3-Phenyl-3-(4-trifluoromethyl-phenoxy)-propylamine 96%
- 5g
- $ 6900.00
- J&W Pharmlab
- (S)-3-Phenyl-3-(4-trifluoromethyl-phenoxy)-propylamine 96%
- 1g
- $ 1880.00
- J&W Pharmlab
- (S)-3-Phenyl-3-(4-trifluoromethyl-phenoxy)-propylamine 96%
- 100mg
- $ 325.00
- J&W Pharmlab
- (S)-3-Phenyl-3-(4-trifluoromethyl-phenoxy)-propylamine 96%
- 50mg
- $ 212.00
- J&W Pharmlab
- (S)-3-Phenyl-3-(4-trifluoromethyl-phenoxy)-propylamine 96%
- 500mg
- $ 998.00
- J&W Pharmlab
- (S)-3-Phenyl-3-(4-trifluoromethyl-phenoxy)-propylamine 96%
- 250mg
- $ 549.00
- American Custom Chemicals Corporation
- (S)-3-PHENYL-3-(4-TRIFLUOROMETHYL-PHENOXY)-PROPYLAMINE 95.00%
- 5MG
- $ 505.78
- Vapor Pressure:5.21E-06mmHg at 25°C
- Refractive Index:1.525
- Boiling Point:381.1°C at 760 mmHg
- Flash Point:184.3°C
- PSA:35.25000
- Density:1.204g/cm3
- LogP:4.87460
- Storage Temp.:Hygroscopic, -20?C Freezer, Under Inert Atmosphere
- XLogP3:3.5
- Hydrogen Bond Donor Count:1
- Hydrogen Bond Acceptor Count:5
- Rotatable Bond Count:5
- Exact Mass:295.11839862
- Heavy Atom Count:21
- Complexity:294
- Purity/Quality:
- 97% *data from raw suppliers
(S)-Norfluoxetine *data from reagent suppliers
- Pictogram(s):
- Hazard Codes:
- MSDS Files:
- SDS file from LookChem
- Canonical SMILES:C1=CC=C(C=C1)C(CCN)OC2=CC=C(C=C2)C(F)(F)F
- Isomeric SMILES:C1=CC=C(C=C1)[C@H](CCN)OC2=CC=C(C=C2)C(F)(F)F
- UsesA metabolite of Fluoxetine (F597100), a selective serotonin reuptake inhibitor which is used as an antidepressant. (S)-Norfluoxetine is a labelled Fluoxetine (F597100) derivative,it is a stereoselective inhibitorof serotonin re-uptake and phosphodiesterase. It is used as a potential antidepressants. A labelled metabolite of Fluoxetine, a selective serotonin reuptake inhibitor which is used as an antidepressant. A metabolite of Fluoxetine, a selective serotonin reuptake inhibitor which is used as an antidepressant.
There total 16 articles about Seproxetine which guide to synthetic route it. The literature collected by LookChem mainly comes from the sharing of users and the free literature resources found by Internet computing technology. We keep the original model of the professional version of literature to make it easier and faster for users to retrieve and use. At the same time, we analyze and calculate the most feasible synthesis route with the highest yield for your reference as below:
Reference yield: 96.0%
- Guidance literature:
-
With dimethylsulfide borane complex; In tetrahydrofuran; at 65 ℃; for 5h;
Reference yield: 65.0%
- Guidance literature:
-
With sodium hydride; In dimethyl sulfoxide; at 90 – 100 ℃; for 1h;
Reference yield:
- Guidance literature:
-
With sodium hydride; In N,N-dimethyl-formamide; at 20 ℃; Resolution of racemate;
-
Buy (S)-3-PHENYL-3- (4-TRIFLUOROMETHYL-PHENOXY) …
Seproxetine, also known as S-norfluoxetine, is a selective serotonin reuptake inhibitor (SSRI). Although its development as an antidepressant was discontinued due to safety concerns, research suggests it …
-
(S)-Norfluoxetine TR-N681095 | CymitQuimica
Seproxetine [INN] Unii-25Co3X0R31 CAS: 126924-38-7 Description: Applications A metabolite of Fluoxetine (F597100), a selective serotonin reuptake inhibitor which is used as an antidepressant. …
-
Seproxetine – LookChem
(S)-Norfluoxetine is a labelled Fluoxetine (F597100) derivative,it is a stereoselective inhibitorof serotonin re-uptake and phosphodiesterase. It is used as a potential antidepressants.
-
CAS RN 126924-38-7 | Fisher Scientific
CAS: 126924-38-7 Chemical Name or Material: (S)-Norfluoxetine Formula Weight: 295.1184 InChI Formula: InChI=1S/C16H16F3NO/c17-16 (18,19)13-6-8-14 (9-7-13)21-15 (10-11-20)12-4-2-1-3-5-12/h1 …
-
126924-38-7 | CAS DataBase – ChemicalBook
ChemicalBook provide information on the 126924-38-7: structure, uses, msds, molecular formula, cas, and suppliers.
-
126924-38-7, (S)-Norfluoxetine Formula – ECHEMI
Get to learn about properities of (S)-Norfluoxetine. The overall knowledge and encyclopedia of (S)-Norfluoxetine covering characteristics, safety, (S)-Norfluoxetine usage, MSDS/SDS and more.
-
(3S)-3-Phenyl-3- [4- (trifluoromethyl)phenoxy]-1-propanamine – CAS …
Explore detailed data on CAS 126924-38-7: (3S)-3-Phenyl-3- [4- (trifluoromethyl)phenoxy]-1-propanamine. Learn about its chemical properties, applications, suppliers, MSDS, and sourcing …
-
CAS: 126924-38-7 (S)-Norfluoxetine | Product Detail Information …
Molecular Formula: 295.30 Formula Weight: C16H16F3NO Inchl Key: WIQRCHMSJFFONW-HNNXBMFYSA-N Inchl: InChI=1S/C16H16F3NO/c17-16 (18,19)13-6-8-14 (9-7-13)21-15 (10-11-20 …
-
(S)-3-Phenyl-3- (4-trifluoromethyl-phenoxy) …
Product Detail (S)-3-Phenyl-3- (4-trifluoromethyl-phenoxy)-propylamine Catalog ID 20R0262 CAS# 126924-38-7 MDL# MFCD00872148 Name (S)-3-Phenyl-3- (4 …
-
(S)-Norfluoxetine (CAS 126924-38-7) – SCBT – Santa Cruz …
(S)-Norfluoxetine, also known as Seproxetine, is a selective serotonin reuptake inhibitor (SSRI) and a metabolite of Fluoxetine. It is a medication used for the treatment of major depression, anxiety …





Reviews
There are no reviews yet.