Buy Muscimol (agarin) Cas 2763-96-4
Buy Muscimol (agarin) Cas 2763-96-4
Muscimol, also known as agarin, pantherine, or pyroibotenic acid, is a GABAA receptor agonist with sedative and hallucinogenic effects and the principal psychoactive constituent of Amanita mushrooms such as Amanita muscaria (fly agaric) and Amanita pantherina (panther cap).[13][1][14][15][16] It is a 3-hydroxyisoxazole alkaloid and is closely related structurally to the neurotransmitter γ-aminobutyric acid (GABA).[14][1][2] The compound is widely used as a ligand and agonist of the GABAA receptor in scientific research.[14][1] Muscimol is typically taken orally, but may also be smoked.[1][12][2][3] Peak effects occur after 1 to 3 hours orally[3][7][9] and its duration is 4 to 8 hours but up to 24 hours.[3][2][11]
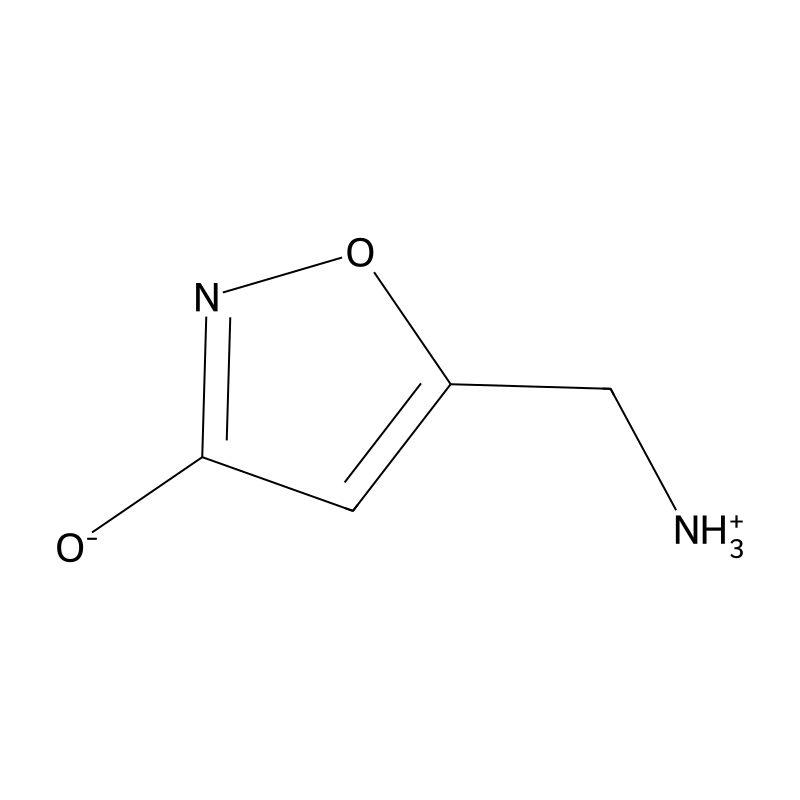
The effects of muscimol in humans include central depression, sedation, sleep, cognitive and motor impairment, hallucinations, perceptual distortion, and muscle twitching, among others.[17][12][18][8] Muscimol acts as a potent GABAA receptor full agonist.[14][1][19] It is also a potent GABAA-ρ receptor partial agonist and a weak GABA reuptake inhibitor.[14][1][2] The drug is inactive at the GABAB receptor but is a substrate of GABA transaminase (GABA-T).[14][12][2] Muscimol mostly exerts its effects via GABAA receptor activation.[12] It is very different from drugs like benzodiazepines and barbiturates as it is an orthosteric agonist of the GABAA receptor rather than an allosteric modulator.[1][20] Unlike GABA, muscimol crosses the blood–brain barrier and hence is centrally active.[20][15][12] Muscimol, which is also known chemically as 5-aminomethylisoxazol-3-ol, is a conformationally restrained analogue of GABA.[14][2] The related compound and Amanita spp. constituent ibotenic acid is a prodrug of muscimol.[1][15][2]
Muscimol was first isolated from Amanita muscaria and hence was discovered in 1964.[14][1][16] It has been limitedly clinically studied as a potential pharmaceutical drug for a number of uses, such as treatment of epilepsy.[1][21][22][23] In addition, analogues and derivatives of muscimol, such as the selective GABAA receptor agonist gaboxadol (THIP; LU-2-030) and the selective GABA reuptake inhibitor tiagabine (Gabitril), have been developed as pharmaceutical drugs.[14][24][2][25] Muscimol and Amanita muscaria mushrooms have rarely been used as recreational drugs historically.[3] By the mid-2020s however, use of these substances, including recreational use for hallucinogenic effects and microdosing for claimed therapeutic benefits, has become increasingly prominent.[1][26][5][27] The most commonly cited therapeutic reason for their use is to improve sleep.[26] Muscimol is not a controlled substance and is unregulated in most of the world, including in most of the United States and Europe.[1][4][5]
Natural occurrence

The main natural sources of muscimol are fungi of the genus Amanita, such as Amanita muscaria (fly agaric) and Amanita pantherina (panther cap). It is produced in the mushrooms along with muscarine (which is present in trace amounts and it is not active), muscazone, and ibotenic acid.[28][15] In Amanita muscaria, the layer just below the skin of the cap contains the highest amount of muscimol, and is therefore the most psychoactive portion.[29]
Use and effects
The properties and effects of muscimol in humans have been limitedly assessed in clinical studies.[12][7][18][8] It has been assessed in these studies at doses of 5 to 15 mg orally.[12] The oral threshold dose of muscimol is approximately 6 mg,[12][30] while the psychoactive dose range has been said to range from approximately 8 to 15 mg.[1][2][11] As little as 1 g of dried Amanita muscaria button may contain this amount of muscimol, although the potency varies greatly among mushrooms.[31] According to Jonathan Ott, a 15 mg dose is “psychoptic” while a 20 mg dose is “visionary”.[3] The onset of action of muscimol, via isolated muscimol or Amanita muscaria consumption orally, is between 30 minutes and 2 hours,[2][8] with peak effects occurring after 1 to 3 hours.[3][7][9] The duration is 4 to 8 hours, but some effects may persist for up to 24 hours.[3][2][10][11][7][9] In one publication, the effects of muscimol were described as follows:[18][8]
- “Waser (1967) describes the effects of self-administration of 10–15 mg. of muscimol as ‘. . . intense hallucinations as with LSD were missing . . . there resulted considerable disturbances of psychic functions, such as orientation in space and time, visual perception, process of thinking, speech, and some new psychic phenomena of illusions and echo pictures’. Higher doses tended to produce severe intoxication in man, with painful muscular twitching, considerable agitation, and vivid hallucinations.”[18][8]
The effects of muscimol in humans in different studies have been found to include sedation, dizziness, incoordination, relaxation, reduced anxiety, mood improvement, sleep, rich dreaming, difficulty speaking, impaired attention, focus, and concentration, impaired learning, confusion, loss of appetite, stimulation, agitation, hallucinogenic effects, echo-like pseudohallucinations (visual and auditory, vivid hallucinations, psychosis, and delirium.[12][1][2][18] At higher doses, coma, seizures, and death can occur.[2][1] Physical effects of muscimol can include muscle twitching, flushing, slightly increased blood pressure, nausea, vomiting, abdominal pain, and diarrhea, among others.[12][2][1][17] After-effects have been reported to include fatigue, inactivity, and headache and migraine.[12] Muscimol can increase prolactin and growth hormone levels. Buy Muscimol (agarin) Cas 2763-96-4, Buy Muscimol (agarin) Cas 2763-96-4, Buy Muscimol (agarin) Cas 2763-96-4, Buy Muscimol (agarin) Cas 2763-96-4, Buy Muscimol (agarin) Cas 2763-96-4, Buy Muscimol (agarin) Cas 2763-96-4, Buy Muscimol (agarin) Cas 2763-96-4, Buy Muscimol (agarin) Cas 2763-96-4, Buy Muscimol (agarin) Cas 2763-96-4, Buy Muscimol (agarin) Cas 2763-96-4, Buy Muscimol (agarin) Cas 2763-96-4, Buy Muscimol (agarin) Cas 2763-96-4, Buy Muscimol (agarin) Cas 2763-96-4
Buy Muscimol (agarin) Cas 2763-96-4
Muscimol
Content Navigation
CAS Number
Product Name
IUPAC Name
Molecular Formula
Molecular Weight
InChI
InChI Key
SMILES
solubility
Synonyms
Canonical SMILES
Analgesic Properties:
Muscimol’s ability to activate GABA receptors has made it a promising candidate for pain management. Studies suggest it might be more effective than GABA itself in reducing neuropathic pain caused by nerve injuries []. This effect is believed to be due to its ability to decrease neuronal excitability in pain pathways. Additionally, muscimol’s anti-inflammatory and antioxidant properties may contribute to its analgesic effects [].
Exploration in Anxiety and Sleep Disorders:
Muscimol’s GABAergic action could also translate to benefits for anxiety and sleep disorders. GABA is the brain’s primary inhibitory neurotransmitter, and its deficiency is linked to these conditions. Early research suggests muscimol might help manage stress and anxiety []. However, more studies are needed to confirm this and determine its safety and efficacy for these specific uses.
Mechanism of Action:
Understanding how muscimol interacts with the nervous system is crucial for developing safe and targeted therapies. Recent research suggests it directly activates a specific potassium channel called TREK-2, which is expressed in GABAergic neurons []. This activation helps regulate neuronal activity and may contribute to muscimol’s therapeutic effects.
Muscimol is a psychoactive compound primarily found in the Amanita muscaria mushroom, commonly known as the Fly Agaric. It is an isoxazole derivative, recognized for its unique structure and potent effects on the central nervous system. The chemical formula of muscimol is C₄H₆N₂O₂, with a molecular weight of 114.10 g/mol. Muscimol appears as a white crystalline powder and is highly soluble in water, with a melting point ranging from 175 to 177 °C .
Muscimol acts mainly as an agonist for GABA-A receptors, mimicking the inhibitory neurotransmitter gamma-aminobutyric acid. This activation leads to the opening of chloride channels, resulting in hyperpolarization of neurons and decreased neuronal excitability. Such properties make muscimol significant for studying GABAergic mechanisms in various physiological and pathological states .
- Muscimol acts as a potent and selective agonist for the GABAA receptor, the main inhibitory neurotransmitter receptor in the brain []. By mimicking the effects of GABA, muscimol reduces neuronal activity, leading to its sedative and depressant effects [].
- The hallucinogenic properties of muscimol are less understood but may be due to its interaction with other brain receptor systems [].
- Muscimol ingestion can be toxic, causing a range of unpleasant symptoms like nausea, vomiting, dizziness, and hallucinations []. In severe cases, coma and even death can occur.
- Due to its unpredictable effects and potential dangers, self-experimentation with muscimol is strongly discouraged.
- Muscimol is not currently a controlled substance in most countries; however, laws and regulations regarding its possession and use can vary by jurisdiction.
Please note:
- The information provided is for scientific research purposes only and should not be interpreted as medical advice.
- Muscimol ingestion can be dangerous, and consulting a medical professional is crucial if you suspect poisoning.
Muscimol exhibits significant biological activity, primarily through its interaction with GABA receptors. As a potent GABA-A receptor agonist and partial agonist at GABA-C receptors, muscimol influences neuronal activity by enhancing inhibitory signals within the brain. This mechanism results in sedative effects, relaxation, and altered states of consciousness .
In addition to its psychoactive effects, muscimol has been studied for its potential therapeutic applications, including anxiety reduction and neuroprotective properties. Its ability to modulate neurotransmitter systems makes it a valuable tool for investigating synaptic transmission and neural circuit dynamics .
The synthesis of muscimol can be achieved through various methods:
- Gagneux Method (1965):
- Step 1: Reflux of 3-bromo-5-aminomethyl-isoxazole with methanol and potassium hydroxide.
- Step 2: Hydrolysis with hydrochloric acid.
- McCarry Method:
- Step 1: Formation of lithium acetylide from propargyl chloride.
- Step 2: Reaction with ethyl chloroformate.
- Step 3: Hydrolysis and further reactions to yield muscimol.
- Alternative Methods:
Muscimol has several applications across various fields:
- Psychoactive Research: Used in studies exploring altered states of consciousness and relaxation.
- Neuroscience: A valuable compound for investigating GABAergic neurotransmission.
- Therapeutic Potential: Investigated for potential applications in treating anxiety disorders and neurodegenerative diseases due to its neuroprotective effects .
- Cultural Uses: Historically utilized in shamanistic practices and traditional medicine due to its psychoactive properties.
Research on muscimol has highlighted its interactions with various neurotransmitter systems:
- GABAergic System: Muscimol’s primary action is through GABA-A receptor activation, leading to enhanced inhibitory signaling in the brain.
- Comparison with Ibotenic Acid: Ibotenic acid, another compound found in Amanita muscaria, acts as a pro-drug to muscimol, converting into it after ingestion. While ibotenic acid activates glutamate receptors leading to excitatory effects, muscimol’s action is predominantly inhibitory .
- Potential Adverse Effects: High doses or unprocessed forms of Amanita muscaria can lead to confusion or delirium due to ibotenic acid before conversion occurs .
Muscimol shares similarities with several other compounds found in Amanita mushrooms or related species. Here are some notable comparisons:
| Compound | Structure Type | Primary Action | Unique Features |
|---|---|---|---|
| Ibotenic Acid | Isoxazole | Glutamate receptor agonist | Neurotoxic; converts to muscimol after ingestion |
| Muscarine | Alkaloid | Acetylcholine receptor agonist | Affects parasympathetic nervous system |
| Psilocybin | Tryptamine | Serotonin receptor agonist | Classic psychedelic; different mechanism than muscimol |
| Baclofen | GABA derivative | GABA-B receptor agonist | Primarily used as a muscle relaxant |
Muscimol’s uniqueness lies in its specific action on GABA receptors compared to the excitatory effects of ibotenic acid or the distinct mechanisms of other psychedelics like psilocybin .
The evolution of muscimol synthesis methodologies reflects the progressive refinement of organic synthetic strategies over nearly eight decades. The earliest practical approach emerged from the work of Gagneux and colleagues in 1945, establishing the dibromoformaldoxime route as the foundational methodology for muscimol synthesis [5] [6].
Early Methodologies (1945-1980)
The dibromoformaldoxime route represented the first scalable approach to muscimol synthesis, utilizing a four-step sequence beginning with the preparation of dibromoformaldoxime from glyoxylic acid and hydroxylamine sulfate [5]. This methodology employed a 1,3-dipolar cycloaddition strategy where bromonitrile oxide, generated in situ from dibromoformaldoxime, reacted with propargylamine derivatives to construct the isoxazole ring system [6]. Despite its historical significance, this approach suffered from low overall yields of approximately 35% and required careful control of reaction conditions to minimize side reactions [5].
The synthesis proceeded through the formation of 5-chloromethyl-3-bromo-isoxazole as a key intermediate, which was subsequently converted to 5-aminomethyl-3-bromoisoxazole through nucleophilic substitution with ammonia [5]. The final steps involved methanolysis to introduce the hydroxyl group at the 3-position, followed by demethylation to yield muscimol [6]. A significant limitation of this route was the problematic conversion of the chloromethyl intermediate to the aminomethyl derivative, which proceeded in disappointingly low yields of 16% [5].
Mechanistic Refinements (1970s-1980s)
The period from 1970 to 1989 witnessed substantial improvements in synthetic methodology, particularly through the contributions of Krogsgaard-Larsen and McCarry [7] [8]. Krogsgaard-Larsen developed improved synthetic sequences that addressed some of the yield limitations of earlier approaches, while McCarry introduced the propargyl chloride route in 1981 [8].
The McCarry synthesis represented a significant advancement by reducing the synthetic sequence to three steps from propargyl chloride [8]. This approach utilized the lithium acetylide of propargyl chloride, which was treated with ethyl chloroformate to afford ethyl 4-chlorotetrolate [8]. The subsequent cyclization with basic hydroxylamine under carefully controlled pH conditions (8.5-9.0) yielded 5-chloromethyl-3-hydroxyisoxazole in 41% yield [8]. The final aminolysis step with methanolic ammonia provided muscimol in 65% yield, resulting in an overall yield of 18.2% [8].
Advanced Dipolar Cycloaddition Strategies
The work of Chiarino, Napoletano, and Sala in 1987 introduced a refined 1,3-dipolar cycloaddition approach that achieved regiospecific control in the formation of the isoxazole ring [6]. This methodology utilized N-dichloroacetylpropargylamine as the dipolarophile in combination with bromonitrile oxide generated from dibromoformaldoxime [6]. The use of potassium bicarbonate as a weak base in heterogeneous phase minimized the tendency of the nitrile oxide toward dimerization, a significant side reaction that had plagued earlier approaches [6].
The improved protocol achieved 80% yield for the formation of 5-aminomethyl-3-bromoisoxazole and demonstrated superior regioselectivity compared to previous methods [6]. The overall yield of 35% starting from dibromoformaldoxime represented a substantial improvement over earlier dipolar cycloaddition approaches [6]. However, despite these advances, the method still required multiple steps and specialized handling of hazardous intermediates.
Modern Catalytic Strategies for Isoxazole Ring Formation
Contemporary approaches to isoxazole synthesis have embraced catalytic methodologies that offer enhanced efficiency, selectivity, and environmental compatibility. These modern strategies have revolutionized the field by introducing transition metal catalysis, organocatalysis, and photocatalytic processes [9] [10] [11].
Transition Metal-Catalyzed Approaches
Modern catalytic strategies for isoxazole ring formation have increasingly focused on copper-catalyzed azide-alkyne cycloaddition reactions and related methodologies [12] [11]. These approaches offer several advantages over traditional methods, including improved functional group tolerance, mild reaction conditions, and high regioselectivity [13].
Copper-catalyzed cycloaddition reactions have been particularly successful in constructing isoxazole rings through the reaction of nitrile oxides with alkynes [12]. The use of copper oxide nanoparticles as catalysts has enabled mechanochemical conditions that eliminate the need for solvents while maintaining high yields and selectivity [11]. This approach represents a significant advancement in terms of both efficiency and environmental impact.
Recent developments have also explored the use of hypervalent iodine reagents as catalysts for intramolecular oxidative cycloaddition reactions [10]. These methodologies enable the efficient formation of condensed isoxazole derivatives through the cyclization of alkyne- or alkene-tethered aldoximes [10]. The use of 2-iodobenzoic acid as a catalyst in combination with m-chloroperbenzoic acid has proven particularly effective, achieving yields up to 94% under mild conditions [10].
Organocatalytic Methodologies
Organocatalytic approaches have emerged as attractive alternatives to transition metal catalysis, offering advantages in terms of cost, toxicity, and sustainability [9] [13]. Modified β-cyclodextrin catalysts have shown remarkable effectiveness in promoting the formation of isoxazole derivatives through multicomponent reactions [12].
The use of immobilized copper in metformin-functionalized β-cyclodextrin has demonstrated exceptional reusability, maintaining catalytic activity even after seven consecutive runs [12]. This approach combines the benefits of heterogeneous catalysis with the environmental advantages of organocatalysis, representing a significant step toward sustainable synthetic methodologies.
Photocatalytic Strategies
Photochemical approaches to isoxazole synthesis have gained considerable attention due to their ability to operate under mild conditions with minimal waste generation [14] [15]. Continuous flow photochemical processes have been developed for the synthesis of isoxazole derivatives, offering precise control over reaction parameters and improved safety profiles [15].
The use of organic photoredox catalysts in continuous flow systems has enabled the efficient synthesis of 3-methyl-4-arylmethylene isoxazole-5(4H)-ones [15]. These methodologies typically employ readily available starting materials and operate under ambient conditions, making them highly attractive for large-scale applications [15].
Recent advances in LED technology have further enhanced the applicability of photocatalytic methods by providing access to specific wavelengths of light that can selectively activate particular functional groups [14]. This selectivity is crucial for achieving high yields and minimizing side reactions in complex synthetic sequences.
Optimization of Hydroxamic Acid Route
The hydroxamic acid route, first reported in 2006, represents the most efficient synthetic approach to muscimol developed to date [5]. This methodology achieves muscimol synthesis in only two steps with an overall yield of 56-64%, representing a significant improvement over all previous approaches [5].
Route Development and Mechanistic Insights
The hydroxamic acid route begins with the reaction of dimethyl acetylenedicarboxylate with hydroxylamine sulfate under basic conditions to form 3-hydroxy-5-isoxazole hydroxamic acid [5]. This initial cyclization proceeds through the nucleophilic attack of hydroxylamine on the triple bond, followed by intramolecular cyclization to form the isoxazole ring [5]. The reaction requires careful pH control, with optimal conditions involving sodium hydroxide in aqueous solution maintained below 5°C during the addition of the acetylenic ester [5].
The key innovation of this route lies in the use of hydroxamic acid functionality as both a synthetic intermediate and a directing group for subsequent transformations [16]. Hydroxamic acids are known to be potent metal-binding groups capable of coordinating with various metal centers, which facilitates selective reduction reactions .
Muscimol

Content Navigation
CAS Number
Product Name
IUPAC Name
Molecular Formula
Molecular Weight
InChI
InChI Key
SMILES
solubility
Synonyms
Canonical SMILES
Analgesic Properties:
Muscimol’s ability to activate GABA receptors has made it a promising candidate for pain management. Studies suggest it might be more effective than GABA itself in reducing neuropathic pain caused by nerve injuries []. This effect is believed to be due to its ability to decrease neuronal excitability in pain pathways. Additionally, muscimol’s anti-inflammatory and antioxidant properties may contribute to its analgesic effects [].
Exploration in Anxiety and Sleep Disorders:
Muscimol’s GABAergic action could also translate to benefits for anxiety and sleep disorders. GABA is the brain’s primary inhibitory neurotransmitter, and its deficiency is linked to these conditions. Early research suggests muscimol might help manage stress and anxiety []. However, more studies are needed to confirm this and determine its safety and efficacy for these specific uses.
Mechanism of Action:
Understanding how muscimol interacts with the nervous system is crucial for developing safe and targeted therapies. Recent research suggests it directly activates a specific potassium channel called TREK-2, which is expressed in GABAergic neurons []. This activation helps regulate neuronal activity and may contribute to muscimol’s therapeutic effects.
Muscimol is a psychoactive compound primarily found in the Amanita muscaria mushroom, commonly known as the Fly Agaric. It is an isoxazole derivative, recognized for its unique structure and potent effects on the central nervous system. The chemical formula of muscimol is C₄H₆N₂O₂, with a molecular weight of 114.10 g/mol. Muscimol appears as a white crystalline powder and is highly soluble in water, with a melting point ranging from 175 to 177 °C .
Muscimol acts mainly as an agonist for GABA-A receptors, mimicking the inhibitory neurotransmitter gamma-aminobutyric acid. This activation leads to the opening of chloride channels, resulting in hyperpolarization of neurons and decreased neuronal excitability. Such properties make muscimol significant for studying GABAergic mechanisms in various physiological and pathological states .
- Muscimol acts as a potent and selective agonist for the GABAA receptor, the main inhibitory neurotransmitter receptor in the brain []. By mimicking the effects of GABA, muscimol reduces neuronal activity, leading to its sedative and depressant effects [].
- The hallucinogenic properties of muscimol are less understood but may be due to its interaction with other brain receptor systems [].
- Muscimol ingestion can be toxic, causing a range of unpleasant symptoms like nausea, vomiting, dizziness, and hallucinations []. In severe cases, coma and even death can occur.
- Due to its unpredictable effects and potential dangers, self-experimentation with muscimol is strongly discouraged.
- Muscimol is not currently a controlled substance in most countries; however, laws and regulations regarding its possession and use can vary by jurisdiction.
Please note:
- The information provided is for scientific research purposes only and should not be interpreted as medical advice.
- Muscimol ingestion can be dangerous, and consulting a medical professional is crucial if you suspect poisoning.
Muscimol exhibits significant biological activity, primarily through its interaction with GABA receptors. As a potent GABA-A receptor agonist and partial agonist at GABA-C receptors, muscimol influences neuronal activity by enhancing inhibitory signals within the brain. This mechanism results in sedative effects, relaxation, and altered states of consciousness .
In addition to its psychoactive effects, muscimol has been studied for its potential therapeutic applications, including anxiety reduction and neuroprotective properties. Its ability to modulate neurotransmitter systems makes it a valuable tool for investigating synaptic transmission and neural circuit dynamics .
The synthesis of muscimol can be achieved through various methods:
- Gagneux Method (1965):
- Step 1: Reflux of 3-bromo-5-aminomethyl-isoxazole with methanol and potassium hydroxide.
- Step 2: Hydrolysis with hydrochloric acid.
- McCarry Method:
- Step 1: Formation of lithium acetylide from propargyl chloride.
- Step 2: Reaction with ethyl chloroformate.
- Step 3: Hydrolysis and further reactions to yield muscimol.
- Alternative Methods:
Muscimol has several applications across various fields:
- Psychoactive Research: Used in studies exploring altered states of consciousness and relaxation.
- Neuroscience: A valuable compound for investigating GABAergic neurotransmission.
- Therapeutic Potential: Investigated for potential applications in treating anxiety disorders and neurodegenerative diseases due to its neuroprotective effects .
- Cultural Uses: Historically utilized in shamanistic practices and traditional medicine due to its psychoactive properties.
Research on muscimol has highlighted its interactions with various neurotransmitter systems:
- GABAergic System: Muscimol’s primary action is through GABA-A receptor activation, leading to enhanced inhibitory signaling in the brain.
- Comparison with Ibotenic Acid: Ibotenic acid, another compound found in Amanita muscaria, acts as a pro-drug to muscimol, converting into it after ingestion. While ibotenic acid activates glutamate receptors leading to excitatory effects, muscimol’s action is predominantly inhibitory .
- Potential Adverse Effects: High doses or unprocessed forms of Amanita muscaria can lead to confusion or delirium due to ibotenic acid before conversion occurs .
Muscimol shares similarities with several other compounds found in Amanita mushrooms or related species. Here are some notable comparisons:
| Compound | Structure Type | Primary Action | Unique Features |
|---|---|---|---|
| Ibotenic Acid | Isoxazole | Glutamate receptor agonist | Neurotoxic; converts to muscimol after ingestion |
| Muscarine | Alkaloid | Acetylcholine receptor agonist | Affects parasympathetic nervous system |
| Psilocybin | Tryptamine | Serotonin receptor agonist | Classic psychedelic; different mechanism than muscimol |
| Baclofen | GABA derivative | GABA-B receptor agonist | Primarily used as a muscle relaxant |
Muscimol’s uniqueness lies in its specific action on GABA receptors compared to the excitatory effects of ibotenic acid or the distinct mechanisms of other psychedelics like psilocybin .
The evolution of muscimol synthesis methodologies reflects the progressive refinement of organic synthetic strategies over nearly eight decades. The earliest practical approach emerged from the work of Gagneux and colleagues in 1945, establishing the dibromoformaldoxime route as the foundational methodology for muscimol synthesis [5] [6].
Early Methodologies (1945-1980)
The dibromoformaldoxime route represented the first scalable approach to muscimol synthesis, utilizing a four-step sequence beginning with the preparation of dibromoformaldoxime from glyoxylic acid and hydroxylamine sulfate [5]. This methodology employed a 1,3-dipolar cycloaddition strategy where bromonitrile oxide, generated in situ from dibromoformaldoxime, reacted with propargylamine derivatives to construct the isoxazole ring system [6]. Despite its historical significance, this approach suffered from low overall yields of approximately 35% and required careful control of reaction conditions to minimize side reactions [5].
The synthesis proceeded through the formation of 5-chloromethyl-3-bromo-isoxazole as a key intermediate, which was subsequently converted to 5-aminomethyl-3-bromoisoxazole through nucleophilic substitution with ammonia [5]. The final steps involved methanolysis to introduce the hydroxyl group at the 3-position, followed by demethylation to yield muscimol [6]. A significant limitation of this route was the problematic conversion of the chloromethyl intermediate to the aminomethyl derivative, which proceeded in disappointingly low yields of 16% [5].
Mechanistic Refinements (1970s-1980s)
The period from 1970 to 1989 witnessed substantial improvements in synthetic methodology, particularly through the contributions of Krogsgaard-Larsen and McCarry [7] [8]. Krogsgaard-Larsen developed improved synthetic sequences that addressed some of the yield limitations of earlier approaches, while McCarry introduced the propargyl chloride route in 1981 [8].
The McCarry synthesis represented a significant advancement by reducing the synthetic sequence to three steps from propargyl chloride [8]. This approach utilized the lithium acetylide of propargyl chloride, which was treated with ethyl chloroformate to afford ethyl 4-chlorotetrolate [8]. The subsequent cyclization with basic hydroxylamine under carefully controlled pH conditions (8.5-9.0) yielded 5-chloromethyl-3-hydroxyisoxazole in 41% yield [8]. The final aminolysis step with methanolic ammonia provided muscimol in 65% yield, resulting in an overall yield of 18.2% [8].
Advanced Dipolar Cycloaddition Strategies
The work of Chiarino, Napoletano, and Sala in 1987 introduced a refined 1,3-dipolar cycloaddition approach that achieved regiospecific control in the formation of the isoxazole ring [6]. This methodology utilized N-dichloroacetylpropargylamine as the dipolarophile in combination with bromonitrile oxide generated from dibromoformaldoxime [6]. The use of potassium bicarbonate as a weak base in heterogeneous phase minimized the tendency of the nitrile oxide toward dimerization, a significant side reaction that had plagued earlier approaches [6].
The improved protocol achieved 80% yield for the formation of 5-aminomethyl-3-bromoisoxazole and demonstrated superior regioselectivity compared to previous methods [6]. The overall yield of 35% starting from dibromoformaldoxime represented a substantial improvement over earlier dipolar cycloaddition approaches [6]. However, despite these advances, the method still required multiple steps and specialized handling of hazardous intermediates.
Modern Catalytic Strategies for Isoxazole Ring Formation
Contemporary approaches to isoxazole synthesis have embraced catalytic methodologies that offer enhanced efficiency, selectivity, and environmental compatibility. These modern strategies have revolutionized the field by introducing transition metal catalysis, organocatalysis, and photocatalytic processes [9] [10] [11].
Transition Metal-Catalyzed Approaches
Modern catalytic strategies for isoxazole ring formation have increasingly focused on copper-catalyzed azide-alkyne cycloaddition reactions and related methodologies [12] [11]. These approaches offer several advantages over traditional methods, including improved functional group tolerance, mild reaction conditions, and high regioselectivity [13].
Copper-catalyzed cycloaddition reactions have been particularly successful in constructing isoxazole rings through the reaction of nitrile oxides with alkynes [12]. The use of copper oxide nanoparticles as catalysts has enabled mechanochemical conditions that eliminate the need for solvents while maintaining high yields and selectivity [11]. This approach represents a significant advancement in terms of both efficiency and environmental impact.
Recent developments have also explored the use of hypervalent iodine reagents as catalysts for intramolecular oxidative cycloaddition reactions [10]. These methodologies enable the efficient formation of condensed isoxazole derivatives through the cyclization of alkyne- or alkene-tethered aldoximes [10]. The use of 2-iodobenzoic acid as a catalyst in combination with m-chloroperbenzoic acid has proven particularly effective, achieving yields up to 94% under mild conditions [10].
Organocatalytic Methodologies
Organocatalytic approaches have emerged as attractive alternatives to transition metal catalysis, offering advantages in terms of cost, toxicity, and sustainability [9] [13]. Modified β-cyclodextrin catalysts have shown remarkable effectiveness in promoting the formation of isoxazole derivatives through multicomponent reactions [12].
The use of immobilized copper in metformin-functionalized β-cyclodextrin has demonstrated exceptional reusability, maintaining catalytic activity even after seven consecutive runs [12]. This approach combines the benefits of heterogeneous catalysis with the environmental advantages of organocatalysis, representing a significant step toward sustainable synthetic methodologies.
Photocatalytic Strategies
Photochemical approaches to isoxazole synthesis have gained considerable attention due to their ability to operate under mild conditions with minimal waste generation [14] [15]. Continuous flow photochemical processes have been developed for the synthesis of isoxazole derivatives, offering precise control over reaction parameters and improved safety profiles [15].
The use of organic photoredox catalysts in continuous flow systems has enabled the efficient synthesis of 3-methyl-4-arylmethylene isoxazole-5(4H)-ones [15]. These methodologies typically employ readily available starting materials and operate under ambient conditions, making them highly attractive for large-scale applications [15].
Recent advances in LED technology have further enhanced the applicability of photocatalytic methods by providing access to specific wavelengths of light that can selectively activate particular functional groups [14]. This selectivity is crucial for achieving high yields and minimizing side reactions in complex synthetic sequences.
Optimization of Hydroxamic Acid Route
The hydroxamic acid route, first reported in 2006, represents the most efficient synthetic approach to muscimol developed to date [5]. This methodology achieves muscimol synthesis in only two steps with an overall yield of 56-64%, representing a significant improvement over all previous approaches [5].
Route Development and Mechanistic Insights
The hydroxamic acid route begins with the reaction of dimethyl acetylenedicarboxylate with hydroxylamine sulfate under basic conditions to form 3-hydroxy-5-isoxazole hydroxamic acid [5]. This initial cyclization proceeds through the nucleophilic attack of hydroxylamine on the triple bond, followed by intramolecular cyclization to form the isoxazole ring [5]. The reaction requires careful pH control, with optimal conditions involving sodium hydroxide in aqueous solution maintained below 5°C during the addition of the acetylenic ester [5].
The key innovation of this route lies in the use of hydroxamic acid functionality as both a synthetic intermediate and a directing group for subsequent transformations [16]. Hydroxamic acids are known to be potent metal-binding groups capable of coordinating with various metal centers, which facilitates selective reduction reactions [16] [17].
Optimization Studies
Extensive optimization of reaction conditions has led to significant improvements in both yield and practicality of the hydroxamic acid route [5]. The initial cyclization step has been optimized to achieve 56% yield of 3-hydroxy-5-isoxazole hydroxamic acid through careful control of temperature, pH, and reagent stoichiometry [5].
The use of anhydrous ethanol for the extraction and purification of the hydroxamic acid intermediate has proven crucial for achieving high yields in the subsequent reduction step [5]. Alternative solvents such as acetone can be employed, although they require larger volumes due to reduced solubility of the hydroxamic acid [5].
Reduction Methodology
The second step of the hydroxamic acid route involves the reduction of the hydroxamic acid to the corresponding amine using borane dimethylsulfide complex [5]. This reduction proceeds under carefully controlled conditions in dry tetrahydrofuran, requiring strict exclusion of moisture to prevent side reactions [5].
The reduction mechanism involves the initial formation of a borane-hydroxamic acid complex, followed by selective reduction of the carbon-nitrogen double bond [5]. The reaction requires extended heating at reflux for 22 hours to ensure complete conversion [5]. Following the reduction, careful quenching with anhydrous methanol is essential to destroy excess borane and prevent formation of amine-borane complexes [5].
The final workup involves treatment with hydrogen chloride gas to cleave any remaining amine-borane adducts, followed by aqueous workup and ion-exchange chromatography to isolate pure muscimol [5]. This purification strategy ensures removal of all synthetic impurities and provides muscimol in high purity suitable for biological applications [5].
Stereochemical Control in Aminomethyl Group Introduction
The introduction of the aminomethyl group at the 5-position of the isoxazole ring represents one of the most challenging aspects of muscimol synthesis, requiring precise control of both regioselectivity and stereochemistry [18] [19]. This transformation is critical because the
Optimization Studies
Extensive optimization of reaction conditions has led to significant improvements in both yield and practicality of the hydroxamic acid route [5]. The initial cyclization step has been optimized to achieve 56% yield of 3-hydroxy-5-isoxazole hydroxamic acid through careful control of temperature, pH, and reagent stoichiometry [5].
The use of anhydrous ethanol for the extraction and purification of the hydroxamic acid intermediate has proven crucial for achieving high yields in the subsequent reduction step [5]. Alternative solvents such as acetone can be employed, although they require larger volumes due to reduced solubility of the hydroxamic acid [5].
Reduction Methodology
The second step of the hydroxamic acid route involves the reduction of the hydroxamic acid to the corresponding amine using borane dimethylsulfide complex [5]. This reduction proceeds under carefully controlled conditions in dry tetrahydrofuran, requiring strict exclusion of moisture to prevent side reactions [5].
The reduction mechanism involves the initial formation of a borane-hydroxamic acid complex, followed by selective reduction of the carbon-nitrogen double bond [5]. The reaction requires extended heating at reflux for 22 hours to ensure complete conversion [5]. Following the reduction, careful quenching with anhydrous methanol is essential to destroy excess borane and prevent formation of amine-borane complexes [5].
The final workup involves treatment with hydrogen chloride gas to cleave any remaining amine-borane adducts, followed by aqueous workup and ion-exchange chromatography to isolate pure muscimol [5]. This purification strategy ensures removal of all synthetic impurities and provides muscimol in high purity suitable for biological applications [5].
Stereochemical Control in Aminomethyl Group Introduction
The introduction of the aminomethyl group at the 5-position of the isoxazole ring represents one of the most challenging aspects of muscimol synthesis, requiring precise control of both regioselectivity and stereochemistry [18] [19]. This transformation is critical because the
-
Buy Muscimol | 2763-96-4 | >98% – Smolecule
Description Muscimol is a psychoactive compound primarily …
-
Muscimol (Agarin, NSC 333569, Pantherine, CAS Number: 2763-96-4 …
Muscimol is a full GABA A agonist and partial GABA C agonist.
-
Muscimol powder 2763-96-4 – MilliporeSigma
Muscimol is a psychoactive isoxazole component, which is found in Amanita muscaria and related mushrooms. [4] It lowers cerebral glucose metabolism and increases the level of 5 …
-
Muscimol | CAS No. 2763-96-4 | Clearsynth
Buy Muscimol CAS No- 2763-96-4, a high quality product from Clearsynth, a Leading Manufacturer and Exporter of Muscimol accompanied by Certificate of Analysis. Click to check details.
-
CAS No : 2763-96-4 | Product Name : Muscimol | Pharmaffiliates
A potent but toxic structural analogue of g-aminobutyric acid (GABA), with a zwitterionic structure that can cross the blood-brain barrier. The PASL product information for any accuracy or completeness is …
-
Reviews: 5
-
Catalogue number: PA PHY 004486
-
CAS Number: 2763-96-4
-
Chemical name: Muscimol
-
-
Muscimol | CAS:2763-96-4 | GABAA receptor agonist, …
BioCrick is a famous high-purity reference standards manufacturer. Our Muscimol is confirmed by NMR. Order now can get a discount!
-
Muscimol CAS 2763-96-4 – CDMO for Pharm & Bio – Kilobio
Muscimol CAS 2763-96-4 New molecule Package in 1kg/bag to 25kg/drum as per request. Molecule modification on request (gram to kilos scale).
-
Muscimol | GABAA Receptors | Tocris Bioscience
View and buy high purity Muscimol from Tocris Bioscience. Potent GABAA …
-
Muscimol (CRM) | CAS 2763-96-4 | Cayman Chemical | Biomol.com
Buy Muscimol (CRM), CAS: 2763-96-4, item number: Cay38030-1 from Cayman Chemical at Biomol!
-
Muscimol | CAS No- 2763-96-4 | Agarine, Pantherine
Buy high quality Muscimol with CAS No – 2763-96-4 from Chemicea Pharma. Chemicea Pharma is one of the leading manufacturer and exporter of Muscimol



Reviews
There are no reviews yet.