Buy Spiradoline (U-62066) Cas 87151-85-7
Buy Spiradoline (U-62066) Cas 87151-85-7
Spiradoline (U-62066) is a drug which acts as a highly selective κ-opioid agonist.[1] It has analgesic,[2] diuretic,[3] and antitussive effects,[4] and produces subjective effects in animals similar to those of ketazocine and alazocine.[5] The main effect in humans is sedation, along with analgesic and diuretic effects, but significant side effects such as dysphoria and hallucinations have stopped it from being used clinically. Buy Spiradoline (U-62066) Cas 87151-85-7
See also
References
-
Product NameSpiradoline
-
NoteResearch use only, not for human use.
-
Brief DescriptionSpiradoline (U 62066) is a potent, highly selective κ-opioid agonist that has analgesic diuretic and antitussive effects.
-
DescriptionSpiradoline (U 62066) is a potent, highly selective κ-opioid agonist that has analgesic diuretic and antitussive effects.Pain Phase 2 Discontinued.
-
In VitroUsing the patch-clamp method in isolated rat cardiac myocytes, indicated that Spiradoline (15 to 500 μM) produces its antiarrythmic effect via blockade of sodium channels (and at the higher doses also of potassium currents) in myocardial tissue. Thus, Spiradoline reduces the peak sodium current, increased the decay rate of the transient outward potassium current, and reduced the sustained plateau potassium amplitude.
-
In VivoSpiradoline (U-62066; 0.1-0.4 mg/kg; subcutaneous injection; once; Sprague-Dawley rats) treatment dose-dependently reduces social behaviors in non-stressed adults, producing social avoidance at the highest dose tested, while younger animals displays reduced sensitivity to this socially suppressing effect of Spiradoline. In stressed animals, the socially suppressing effects of the Spiradoline are blunted at all ages, with juveniles and adolescents exhibiting increased social preference in response to certain doses of U-62066. Animal Model:Juvenile, adolescent and adult Sprague-Dawley male and female rats exposured to repeated restraint Dosage:0.1 mg/kg, 0.2 mg/kg, 0.3 mg/kg, and 0.4?mg/kg Administration:Subcutaneous injection; once Result:Dose-dependently reduced social behaviors in non-stressed adults, producing social avoidance at the highest dose tested. Buy Spiradoline (U-62066) Cas 87151-85-7
-
SynonymsU 62066
-
PathwayEndocrinology/Hormones
-
TargetOpioid Receptor
-
RecptorOpioid Receptor
-
Research AreaNeurological Disease
-
IndicationPain
Chemical Information
-
CAS Number87151-85-7
-
Formula Weight425.392
-
Molecular FormulaC22H30Cl2N2O2
-
Purity>98% (HPLC)
-
SolubilityIn Vitro:?DMSO : 50 mg/mL (117.54 mM)
-
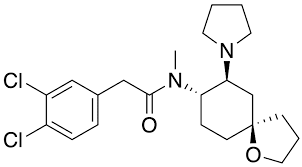
SMILESCN([C@H]1CC[C@@]2(CCCO2)C[C@@H]1N3CCCC3)C(=O)CC4=CC(=C(C=C4)Cl)Cl.CS(=O)(=O)O
-
Chemical Name2-(3,4-dichlorophenyl)-N-methyl-N-[(5R,7S,8S)-7-pyrrolidin-1-yl-1-oxaspiro[4.5]decan-8-yl]acetamide
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
U-62066 is a compound frequently used in research focusing on its binding affinity and action at opioid receptors. In studies pertaining to neuroscience, U-62066 is applied to investigate the modulation of neurotransmitter release and its subsequent effects on neural pathways. This compound is also of interest in examining the role of opioid receptors in the regulation of pain signaling pathways. Additionally, U-62066 is useful for researchers studying the mechanisms of addiction, as it provides a means to understand the complex interactions between neurotransmitter systems and behavior. In biochemistry, U-62066 is employed to study receptor-ligand interactions and to characterize the activation or inhibition of signal transduction processes.
U-62066 (CAS 87151-85-7) References
- Comparison of cardiovascular responses to intra-hippocampal mu, delta and kappa opioid agonists in spontaneously hypertensive rats and isolation-induced hypertensive rats. | Shen, S. and Ingenito, AJ. 1999. J Hypertens. 17: 497-505. PMID: 10404951
- Internalization of mu-opioid receptors in rat spinal cord slices. | Marvizón, JC., et al. 1999. Neuroreport. 10: 2329-34. PMID: 10439458
- Effects of a kappa agonist, spiradoline mesylate (U62,066E), on activation and vaginocervical-stimulation produced analgesia in rats. | Boyle, TJ., et al. 2001. Brain Res Bull. 54: 213-8. PMID: 11275411
- Kappa agonist-induced reduction in dopamine release: site of action and tolerance. | Donzanti, BA., et al. 1992. Res Commun Chem Pathol Pharmacol. 78: 193-210. PMID: 1282270
- Central monoaminergic mechanisms in mice and analgesic activity of spiradoline mesylate, a selective kappa-opioid receptor agonist. | Kunihara, M., et al. 1992. Eur J Pharmacol. 214: 111-8. PMID: 1325350
- Roles of different subtypes of opioid receptors in mediating the ventrolateral orbital cortex opioid-induced inhibition of mirror-neuropathic pain in the rat. | Zhao, M., et al. 2007. Neuroscience. 144: 1486-94. PMID: 17184926
- Mu-opioid receptor in the nucleus submedius: involvement in opioid-induced inhibition of mirror-image allodynia in a rat model of neuropathic pain. | Wang, JY., et al. 2008. Neurochem Res. 33: 2134-41. PMID: 18473169
- Role of mu, delta and kappa opioid receptors in ethanol-reinforced operant responding in infant rats. | Miranda-Morales, RS., et al. 2012. Behav Brain Res. 234: 267-77. PMID: 22789403
- Analgesic activity of spiradoline mesylate (U-62,066E), a kappa opioid agonist in mice. | Kunihara, M., et al. 1989. Life Sci. 45: 1191-8. PMID: 2796604
- Quantitative analysis of effects of kappa-opioid agonists on postischemic hippocampal CA1 neuronal necrosis in gerbils. | Hall, ED. and Pazara, KE. 1988. Stroke. 19: 1008-12. PMID: 2840759
- Cardiovascular depressant effects of the kappa opioid receptor agonists U50488H and spiradoline mesylate. | Hall, ED., et al. 1988. Circ Shock. 26: 409-17. PMID: 2850873
- Effects of spiradoline mesylate, a selective kappa-opioid-receptor agonist, on the central dopamine system with relation to mouse locomotor activity and analgesia. | Kunihara, M., et al. 1993. Jpn J Pharmacol. 62: 223-30. PMID: 8411771
- New degradation product of spiradoline mesylate in aqueous solution: formation of an imidazolidine ring. | Ogata, M., et al. 1993. J Pharm Sci. 82: 91-4. PMID: 8429499
- Gender differences in kappa-opioid modulation of cocaine-induced behavior and NMDA-evoked dopamine release. | Sershen, H., et al. 1998. Brain Res. 801: 67-71. PMID: 9729284
Spiradoline (U-62066), an arylacetamide, is a selective kappa opioid receptor (KOR) agonist with a Ki of 8.6 nM in guinea pig. The Ki values of Spiradoline for μ and δ receptors are 252 nM and 9400 nM, respectively. Spiradoline has potent diuretic, analgesic, antiarrythmic, antitussive, neuroprotective properties and easily penetrates the blood-brain barrier[1][2].
Using the patch-clamp method in isolated rat cardiac myocytes, indicated that Spiradoline (15 to 500 μM) produces its antiarrythmic effect via blockade of sodium channels (and at the higher doses also of potassium currents) in myocardial tissue. Thus, Spiradoline reduces the peak sodium current, increased the decay rate of the transient outward potassium current, and reduced the sustained plateau potassium amplitude[2].
Spiradoline (U-62066; 0.1-0.4 mg/kg; subcutaneous injection; once; Sprague-Dawley rats) treatment dose-dependently reduces social behaviors in non-stressed adults, producing social avoidance at the highest dose tested, while younger animals displays reduced sensitivity to this socially suppressing effect of Spiradoline. In stressed animals, the socially suppressing effects of the Spiradoline are blunted at all ages, with juveniles and adolescents exhibiting increased social preference in response to certain doses of U-62066[1].
[1]. Elena I Varlinskaya, et al. Stress alters social behavior and sensitivity to pharmacological activation of kappa opioid receptors in an age-specific manner in Sprague Dawley rats. Neurobiol Stress. 2018 Sep 11;9:124-132.
[2]. M-L G Wadenberg. A review of the properties of spiradoline: a potent and selective kappa-opioid receptor agonist. CNS Drug Rev. Summer 2003;9(2):187-98.
| Cas No. | 87151-85-7 | SDF | |
| Formula | C22H30Cl2N2O2 | M.Wt | 425.39 |
| Solubility | DMSO : 50 mg/mL (117.54 mM; Need ultrasonic) | Storage | |
| General tips | Please select the appropriate solvent to prepare the stock solution according to the solubility of the product in different solvents; once the solution is prepared, please store it in separate packages to avoid product failure caused by repeated freezing and thawing.Storage method and period of the stock solution: When stored at -80°C, please use it within 6 months; when stored at -20°C, please use it within 1 month. To increase solubility, heat the tube to 37°C and then oscillate in an ultrasonic bath for some time. |
||
| Shipping Condition | Evaluation sample solution: shipped with blue ice. All other sizes available: with RT, or with Blue Ice upon request. | ||
| Prepare stock solution | |||
 |
1 mg | 5 mg | 10 mg |
| 1 mM | 2.3508 mL | 11.7539 mL | 23.5078 mL |
| 5 mM | 470.2 μL | 2.3508 mL | 4.7016 mL |
| 10 mM | 235.1 μL | 1.1754 mL | 2.3508 mL |
| Targets |
Ki: 8.6 nM (κ-opioid receptor in guinea pig), 252 nM (μ-receptor) and 9400 nM (δ-receptor)[2]
|
|---|---|
| ln Vitro |
Employing the patch-clamp technique on isolated rat heart myocytes, it was found that spirol (15–500 μM) inhibits potassium currents and sodium channels in cardiac tissue to create its antiarrhythmic activity. At higher concentrations, it also blocks potassium channels. Consequently, spiradoline decreases the sustained plateau potassium amplitude, increases the decay rate of the transient outward potassium current, and decreases the peak sodium current[2].
|
| ln Vivo |
In non-stressed adults, Spiradoline (U-62066; 0.1-0.4 mg/kg; subcutaneous injection; once; Sprague-Dawley rats) treatment dose-dependently decreases social behaviors, resulting in social avoidance at the highest dose tested. However, in younger animals, Spiradoline’s socially suppressive effect is less pronounced. The Spiradoline’s socially suppressive effects are attenuated in stressed animals at all ages, with juveniles and adolescents responding to specific dosages of U-62066 with greater social preference[1].
|
| Animal Protocol |
Animal/Disease Models: Juvenile, adolescent and adult SD (Sprague-Dawley) male and female rats exposed to repeated restraint[1]
Doses: 0.1 mg/kg, 0.2 mg/kg, 0.3 mg/kg, and 0.4 mg/kg Route of Administration: subcutaneous (sc) injection; once Experimental Results: Dose -dependently decreased social behaviors in non-stressed adults, producing social avoidance at the highest dose tested. |
| References |
|
| Additional Infomation |
Spiradoline has been investigated for the basic science of Bipolar Depression.
|
| Molecular Formula |
C22H30CL2N2O2
|
|---|---|
| Molecular Weight |
425.39
|
| Exact Mass |
424.168
|
| CAS # |
87151-85-7
|
| Related CAS # |
Spiradoline mesylate;87173-97-5
|
| PubChem CID |
55652
|
| Appearance |
Typically exists as solid at room temperature
|
| LogP |
4.498
|
| Hydrogen Bond Donor Count |
0
|
| Hydrogen Bond Acceptor Count |
3
|
| Rotatable Bond Count |
4
|
| Heavy Atom Count |
28
|
| Complexity |
557
|
| Defined Atom Stereocenter Count |
3
|
| SMILES |
N([C@H]1CC[C@]2(OCCC2)C[C@@H]1N1CCCC1)(C)C(=O)CC1C=CC(Cl)=C(Cl)C=1
|
| InChi Key |
NYKCGQQJNVPOLU-ONTIZHBOSA-N
|
| InChi Code |
InChI=1S/C22H30Cl2N2O2/c1-25(21(27)14-16-5-6-17(23)18(24)13-16)19-7-9-22(8-4-12-28-22)15-20(19)26-10-2-3-11-26/h5-6,13,19-20H,2-4,7-12,14-15H2,1H3/t19-,20-,22-/m0/s1
|
| Chemical Name |
2-(3,4-dichlorophenyl)-N-methyl-N-[(5R,7S,8S)-7-pyrrolidin-1-yl-1-oxaspiro[4.5]decan-8-yl]acetamide
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
DMSO: 50 mg/mL (117.54 mM)
|
|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.88 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.88 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. View MoreSolubility in Formulation 3: ≥ 2.5 mg/mL (5.88 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), suspension solution.
(Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3508 mL | 11.7539 mL | 23.5078 mL | |
| 5 mM | 0.4702 mL | 2.3508 mL | 4.7016 mL | |
| 10 mM | 0.2351 mL | 1.1754 mL | 2.3508 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
-
U-62066 | CAS 87151-85-7 | SCBT – Santa Cruz …
U-62066 is a compound frequently used in research focusing on its binding affinity and action at opioid receptors. In studies pertaining to neuroscience, U-62066 is …
-
U-62066 solid 87151-85-7 – MilliporeSigma
Spiradoline (U-62,066E), a selective kappa (kappa) receptor agonist, was examined for actions on the cardiovascular system and on myocardial ionic currents in rats.
-
U-62066 (CAS 87151-85-7) | Glentham Life Sciences
Buy GL0603 – U-62066 (87151-85-7) online from Glentham Life Sciences, a manufacturer and supplier of fine chemicals. View catalogue prices, chemical data, technical specifications and SDS documents.
-
87151-85-7 | Spiradoline | ChemScene
ChemScene Provide CAS 87151-85-7, Spiradoline, U-62066, Formula:C22H30Cl2N2O2, MW:425.3900. We also provide services including custom synthesis, process optimization, commercial production, …
-
Spiradoline (U-62066) | Opioid Receptor | 87151-85-7 | Invivochem
Spiradoline (U-62066), an arylacetamide, is a selective kappa opioid receptor (KOR) agonist/activator with a Ki of 8.6 nM in guinea pigs. The Kis of Spiradoline for μ and δ receptors are 252 nM and 9400 …
-
Spiradoline | CAS NO.:87151-85-7 | GlpBio
Spiradoline (U-62066), an arylacetamide, is a selective kappa opioid receptor …
Spiradoline |CAS:87151-85-7 Probechem Biochemicals
Catalog No.: PC-61886 Not For Human Use, Lab Use Only. Spiradoline (U 62066) is a potent, highly selective κ-opioid agonist that has analgesic diuretic and antitussive effects..
-
U-62066 | CAS 87151-85-7 | AbMole BioScience | U-62066 Price
Highly selective κ opioid receptor agonist; antitussive. Buy U-62066 from AbMole BioScience.
-
Spiradoline | 87151-85-7 | Opioid Receptor | MOLNOVA
Spiradoline (U 62066) is a potent, highly selective κ-opioid agonist that has analgesic diuretic and antitussive effects.
-
U-62066 (SPIRADOLINE MESYLATE) K-OPIOID AGONIST | 87151 …
U-62066 (SPIRADOLINE MESYLATE) K-OPIOID AGONIST (87151-85-7) information like chemical properties,Structure,melting point,boiling point,density,molecular …



Reviews
There are no reviews yet.