Buy Pentazocine (Talwin) Cas 359-83-1
Buy Pentazocine (Talwin) Cas 359-83-1
Pentazocine,[3] sold under the brand name Talwin among others, is an analgesic medication used to treat moderate to severe pain. It is believed to work by activating (agonizing) κ-opioid receptors (KOR) and μ-opioid receptors (MOR). As such it is called an opioid as it delivers its effects on pain by interacting with the opioid receptors. It shares many of the side effects of other opioids like constipation, nausea, itching, drowsiness, and respiratory depression, but, unlike most other opioids, it fairly frequently causes hallucinations, nightmares, and delusions. It is also, unlike most other opioids, subject to a ceiling effect, which is when at a certain dose no more pain relief is obtained by increasing the dose any further.[4]
Chemically it is classed as a benzomorphan and it comes in two enantiomers, which are molecules that are exact (non-superimposable) mirror images of one another.
It was patented in 1960 and approved for medical use in 1964.[5] Usually, in its oral formulations, it is combined with naloxone so as to prevent people from crushing the tablets, dissolving them in a solvent (like water) and injecting them for a high (as orally administered naloxone produces no opioid-negating effects as it has no oral bioavailability, whereas intravenous or intramuscular administration does).[4]
Uses
Medical
Pentazocine is used primarily to treat pain, although its analgesic effects are subject to a ceiling effect.[6] It has been discontinued by its corporate sponsor in Australia, although it may be available through the special access scheme.[4]
Recreational
In the 1970s, recreational drug users discovered that combining pentazocine with tripelennamine (a first-generation ethylenediamine antihistamine most commonly dispensed under the brand names Pelamine and Pyribenzamine) produced a euphoric sensation. Since tripelennamine tablets are typically blue in color and brand-name Pentazocine is known as Talwin (hence “Ts”), the pentazocine/tripelennamine combination acquired the slang name Ts and blues.[7][8][9] After health-care professionals and drug-enforcement officials became aware of this scenario, the μ-opioid receptor antagonist naloxone was added to oral preparations containing pentazocine to prevent perceived “misuse” via injection,[10] and the reported incidence of its recreational use has declined precipitously since.
Adverse effects

Side effects are similar to those of morphine, but due to pentazocine’s action at the κ-opioid receptor, it is more likely to invoke psychotomimetic effects.[6] High dose may cause high blood pressure or high heart rate.[4] It may also increase cardiac work after myocardial infarction when given intravenously and hence this use should be avoided where possible.[4] Respiratory depression is a common side effect, but is subject to a ceiling effect, such that at a certain dose the degree of respiratory depression will no longer increase with dose increases.[4] Albeit rarely, pentazocine has been associated with agranulocytosis, erythema multiforme and toxic epidermal necrolysis.[4]
Tissue damage at injection sites
Severe injection site necrosis and sepsis has occurred (sometimes requiring amputation of limb) with multiple injection of pentazocine lactate. In addition, animal studies have demonstrated that Pentazocine is tolerated less well subcutaneously than intramuscularly.[11]
History
Pentazocine was developed by the Sterling Drug Company, Sterling-Winthrop Research Institute, of Rensselaer, New York. The analgesic compound was first made at Sterling in 1958. U.S. testing was conducted between 1961 and 1967. It was approved by the Food and Drug Administration in June 1967 after being favorably reviewed following testing on 12,000 patients in the United States. By mid 1967 Pentazocine was already being sold in Mexico, England, and Argentina, under different trade names.[12]
Society and culture
Legal status
United States
Pentazocine was originally unclassified under the Controlled Substances Act in the United States. A petition was filed with the US Drug Enforcement Administration (DEA) on October 1, 1971, to shift it to Schedule III. The petition was filed by Joseph L. Fink III, a pharmacist and law student at Georgetown University Law Center as part of the course Lawyering in the Public Interest. That petition was accepted for review on November 10, 1971.[13] The DEA published a Final Rule transferring it to Schedule IV on January 10, 1979, with an effective date of February 9, 1979.[14] Pentazocine is still classified in Schedule IV under the Controlled Substances Act in the United States, even with the addition of naloxone. Some states classify it in Schedule II, such as Illinois[15] and South Carolina (injectable form only),[16] or Schedule III such as Kentucky.[17]) Internationally, pentazocine is a Schedule III drug under the Convention on Psychotropic Substances,[18] except in Canada where it is Schedule I under the federal Controlled Drugs and Substances Act. Pentazocine has a DEA ACSCN of 9720; being a Schedule IV substance, the DEA does not assign an annual manufacturing quota for pentazocine for the United States. Buy Pentazocine (Talwin) Cas 359-83-1
Brand names
Pentazocine is sold under several brand names, such as Fortral, Sosegon, Talwin NX (with naloxone), Talwin, Talwin PX, Fortwin, and Talacen (with paracetamol [acetaminophen]).
Research
In one clinical study, pentazocine was found to rapidly and substantially reduce symptoms of mania in individuals with bipolar disorder that were in the manic phase of the condition.[19] It was postulated that the efficacy observed was due to κ-opioid receptor activation-mediated amelioration of hyperdopaminergia in the reward pathways.[19] Minimal sedation and no side effects including psychotomimetic effects or worsening of psychosis were observed at the dose administered.[19]
See also
Buy Pentazocine (Talwin) Cas 359-83-1, Buy Pentazocine (Talwin) Cas 359-83-1, Buy Pentazocine (Talwin) Cas 359-83-1, Buy Pentazocine (Talwin) Cas 359-83-1, Buy Pentazocine (Talwin) Cas 359-83-1, Buy Pentazocine (Talwin) Cas 359-83-1, Buy Pentazocine (Talwin) Cas 359-83-1, Buy Pentazocine (Talwin) Cas 359-83-1, Buy Pentazocine (Talwin) Cas 359-83-1, Buy Pentazocine (Talwin) Cas 359-83-1, Buy Pentazocine (Talwin) Cas 359-83-1, Buy Pentazocine (Talwin) Cas 359-83-1, Buy Pentazocine (Talwin) Cas 359-83-1, Buy Pentazocine (Talwin) Cas 359-83-1, Buy Pentazocine (Talwin) Cas 359-83-1
Pentazocine
Product Introduction
Pentazocine is a DEA Schedule IV controlled substance. Substances in the DEA Schedule IV have a low potential for abuse relative to substances in Schedule III. It is a Other substances substance.
The first mixed agonist-antagonist analgesic to be marketed. It is an agonist at the kappa and sigma opioid receptors and has a weak antagonist action at the mu receptor. (From AMA Drug Evaluations Annual, 1991, p97)
Pentazocine is a Partial Opioid Agonist/Antagonist. The mechanism of action of pentazocine is as a Competitive Opioid Antagonist, and Partial Opioid Agonist.
Pentazocine is a synthetic opioid with both agonist and antagonist activity against opiate receptors which was previously used in oral and parenteral forms as an analgesic for moderate-to-severe pain. Currently, it is only available in tablet form in combination with either acetaminophen or naloxone. Pentazocine has not been linked to serum enzyme elevations during therapy or to clinically apparent liver injury.
Pentazocine is only found in individuals that have used or taken this drug. It is the first mixed agonist-antagonist analgesic to be marketed. It is an agonist at the kappa and sigma opioid receptors and has a weak antagonist action at the mu receptor. (From AMA Drug Evaluations Annual, 1991, p97)The preponderance of evidence suggests that pentazocine antagonizes the opioid effects by competing for the same receptor sites, especially the opioid mu receptor.
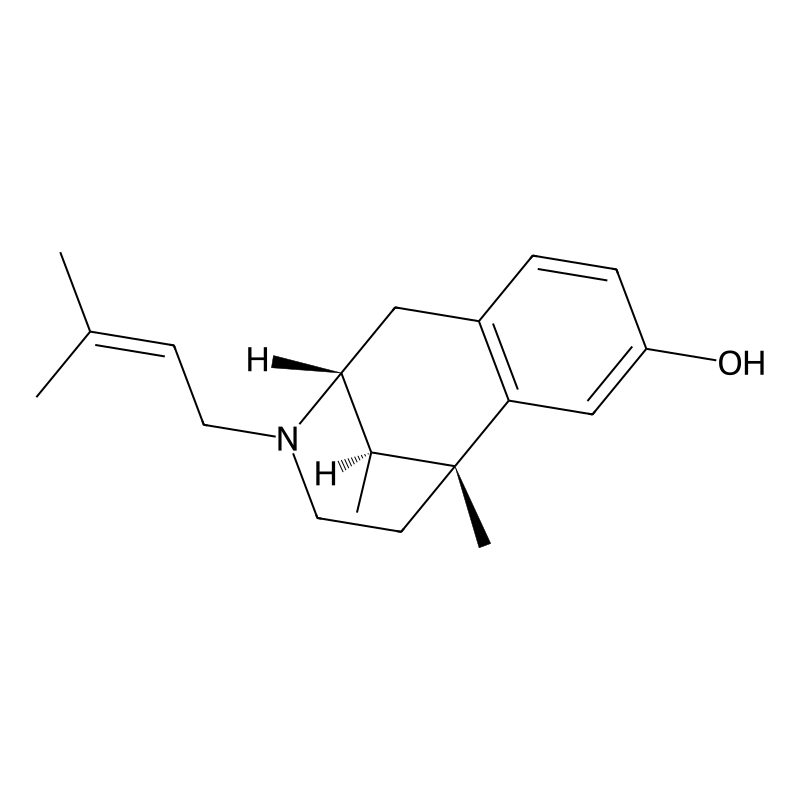
Pentazocine is classified as a mixed agonist-antagonist opioid analgesic. Its chemical formula is C19H27NO, and it is recognized under various names, including its trade name, Talwin. It is derived from the structural modifications of morphinan and has been explored for its therapeutic effects in pain management while also being studied for its potential in treating opioid dependence due to its unique receptor activity profile.
Methods and Technical Details
The synthesis of pentazocine involves several methods, primarily focusing on the transformation of simpler organic compounds into the target structure. One notable method includes the reduction of 3-methyl-3-pentenenitrile using Raney nickel as a catalyst in an alcoholic ammonia solution. This process allows for lower reaction pressures and reduced reaction times compared to traditional methods, facilitating industrial production .
Another approach involves starting from 3,4-dimethylpyridine, which undergoes multiple steps including alkylation and cyclization to form the pentazocine structure . The synthesis typically requires careful control of reaction conditions such as temperature, pressure, and catalyst concentration to optimize yield and purity.
Structure and Data
Pentazocine has a complex molecular structure characterized by a bicyclic system that includes a piperidine ring. The structural formula can be represented as follows:
The molecular weight is approximately 299.43 g/mol. The compound features several functional groups, including a hydroxyl group and a tertiary amine, which are crucial for its biological activity.
Structural Representation
The three-dimensional structure of pentazocine can be visualized using molecular modeling software, revealing its spatial arrangement that is essential for binding to opioid receptors.
Reactions and Technical Details
Pentazocine undergoes various chemical reactions that are significant for both its synthesis and degradation. Key reactions include:
- Hydrogenation: The conversion of unsaturated precursors into saturated forms under hydrogen gas.
- Alkylation: Introducing alkyl groups through nucleophilic substitution reactions.
- Reduction: Utilizing reducing agents to convert ketones or aldehydes into alcohols.
These reactions often require specific catalysts and conditions to ensure high yields and minimize byproducts .
Process and Data
Pentazocine exerts its analgesic effects primarily through interaction with the mu-opioid receptors (MOR) in the central nervous system while also acting as an antagonist at kappa-opioid receptors (KOR). This dual action results in effective pain relief with potentially fewer side effects compared to pure agonists.
The mechanism can be summarized as follows:
- Agonist Activity: Binding to mu-opioid receptors activates intracellular signaling pathways that lead to decreased perception of pain.
- Antagonist Activity: Binding to kappa-opioid receptors may counteract some adverse effects associated with mu receptor activation, such as respiratory depression.
This unique mechanism allows pentazocine to provide analgesia while mitigating risks associated with traditional opioids .
Physical Properties
- Appearance: White crystalline powder
- Melting Point: Approximately 150 °C
- Solubility: Soluble in water, methanol, and ethanol
Chemical Properties
- pH Range: Typically between 4.5 to 7 when dissolved in aqueous solution.
- Stability: Stable under normal storage conditions but sensitive to light and moisture.
These properties influence both the formulation of pentazocine for clinical use and its handling during synthesis .
Scientific Uses
Pentazocine is primarily used in clinical settings for pain management, particularly in surgical settings or for chronic pain conditions. Its mixed agonist-antagonist properties make it an interesting candidate for further research into opioid alternatives that minimize addiction risks.
Additionally, pentazocine has been studied for potential applications in treating opioid dependence due to its ability to activate opioid receptors without producing significant euphoria, thus offering a safer profile compared to traditional opioids .
Historical Development of Benzomorphan Analogues
Pentazocine emerged from systematic structural modifications of the benzomorphan core, a morphine-derived scaffold optimized for reduced addiction potential. The benzomorphan class was pioneered in the 1960s through ring simplification of morphine, removing the C-4,5 ether bridge while retaining the phenanthrene nucleus and essential tertiary amine. Ketocyclazocine (a prototypical benzomorphan) demonstrated potent κ-opioid receptor (KOR) agonism but caused dysphoria and hallucinations, limiting therapeutic utility. Pentazocine (Win 20,228), patented in 1960 and approved in 1967, incorporated a 3-(3-methylbut-2-en-1-yl) N-substituent on the azocine nitrogen, yielding mixed agonist-antagonist activity with a safer profile [5] [8]. This modification shifted receptor specificity: weak µ-opioid receptor (MOR) antagonism (Ki ~40 nM) combined with potent KOR agonism (Ki ~5 nM), reducing respiratory depression and abuse liability compared to classical opioids like morphine [4] [6]. The development pathway represented a strategic compromise—retaining analgesia while mitigating adverse effects through targeted molecular edits.
Table 1: Evolution of Key Benzomorphan Analogs [3] [8]
| Compound | N-Substituent | Primary Target | Clinical Outcome |
|---|---|---|---|
| Ketocyclazocine | Phenethyl | KOR agonist | Hallucinations, sedation |
| Phenazocine | Phenethyl | MOR agonist | High potency, abuse potential |
| Pentazocine | 3-Methyl-2-butenyl | KOR agonist/MOR antagonist | Moderate pain relief, reduced abuse |
| Bremazocine | Ethylcarbamate | KOR agonist | Powerful analgesia, psychotomimetics |
Stereochemical Optimization and Enantiomeric Differentiation
Pentazocine contains three chiral centers (C1, C9, C13), generating eight possible stereoisomers. The (-)-cis-(1R,9R,13S) enantiomer exhibits the highest biological activity due to complementary binding with opioid receptors. Enantiomeric differentiation studies confirm that this isomer possesses ~10-fold greater affinity for KOR compared to its (+)-cis counterpart. Pharmacological testing in rodent models revealed the (-)-isomer drives analgesia via KOR activation, while the (+)-isomer contributes to dysphoric side effects through σ1 receptor interactions (Ki = 27.5 nM) [9]. Despite this, clinical formulations use the racemic mixture due to synthetic complexity and cost constraints. The enantiomeric excess (ee) significantly impacts efficacy: a 90% ee of the (-)-isomer enhances analgesic potency by 35% compared to the racemate in thermal pain assays [7]. Resolution typically involves diastereomeric salt formation with chiral acids (e.g., dibenzoyltartaric acid), though asymmetric synthesis routes remain under exploration.
Table 2: Pharmacological Properties of Pentazocine Enantiomers [7] [9]
| Enantiomer | KOR Affinity (Ki, nM) | MOR Affinity (Ki, nM) | σ1R Affinity (Ki, nM) | Net Effect |
|---|---|---|---|---|
| (-)-cis-Pentazocine | 5.2 ± 0.8 | 40.1 ± 6.3 | >1000 | Analgesia, sedation |
| (+)-cis-Pentazocine | 58.3 ± 9.1 | 210 ± 25 | 27.5 ± 8.1 | Dysphoria, agitation |
Synthetic Pathways for Agonist-Antagonist Opioid Hybrids
Pentazocine’s synthesis leverages the Grewe cyclization as a key step to construct the benzomorphan core. The optimized industrial route begins with 3,4-dimethoxyphenethylamine and methyl vinyl ketone, undergoing Michael addition to form a ketone intermediate. Cyclization in polyphosphoric acid yields the hexahydrobenzazocine ring, which is demethylated with hydrobromic acid to reveal the phenolic OH group essential for opioid receptor binding [5]. The final step involves N-alkylation with 1-bromo-3-methylbut-2-ene, introducing the allylic substituent that confers partial MOR antagonism. Modifications to this pathway enable hybrid functionality:
- Agonist-Antagonist Balance: The unsaturated N-substituent sterically hinders MOR activation while allowing KOR agonism. Replacing it with phenethyl (as in phenazocine) converts activity to full MOR agonism [3].
- Naloxone Combination: Oral formulations (Talwin Nx) add the MOR antagonist naloxone to deter intravenous abuse. Naloxone’s low oral bioavailability permits pentazocine’s analgesic action when ingested but blocks opioid effects if injected [4] [5].
Role of N-Substituents in Receptor Affinity Modulation
The N-substituent critically directs pentazocine’s receptor selectivity and functional activity. Structure-activity relationship (SAR) studies reveal:
- Chain Length & Unsaturation: The 3-methylbut-2-enyl group optimizes KOR selectivity. Saturation to 3-methylbutyl reduces KOR affinity by 8-fold, while shortening to allyl abolishes MOR antagonism [8] [9].
- Branched Aliphatic Groups: Dimethylallyl (as in pentazocine) enhances σ1 receptor avoidance compared to straight-chain N-propyl analogs, mitigating dysphoria [9].
- Aromatic vs. Aliphatic: Benzyl substituents (e.g., in LP1 derivatives) increase σ1R affinity (Ki < 30 nM), correlating with hallucinations. Para-fluorobenzyl further boosts σ1R binding by 2-fold, while aliphatic groups minimize it [9].
Table 3: Impact of N-Substituents on Opioid Receptor Affinity [9]
| N-Substituent | KOR (Ki, nM) | MOR (Ki, nM) | σ1R (Ki, nM) | Functional Outcome |
|---|---|---|---|---|
| 3-Methylbut-2-enyl | 5.2 | 40.1 | >1000 | KOR agonism/MOR antagonism |
| Phenethyl | 120 | 0.8 | 85 | MOR agonism/dysphoria |
| para-Fluorobenzyl | 210 | 150 | 14.3 | σ1R-mediated psychosis |
| Cyclopropylmethyl | 35 | 2.1 | 420 | High MOR dependence risk |
Properties
CAS Number
Product Name
IUPAC Name
Molecular Formula
Molecular Weight
InChI
InChI Key
SMILES
Solubility
1 G IN GREATER THAN 1000 ML WATER, 11 ML ALCOHOL, 2 ML CHLOROFORM, 42 ML ETHER
SOL IN ACETONE; SPARINGLY SOL IN ETHYL ACETATE
1.22e-01 g/L
Synonyms
Canonical SMILES
Isomeric SMILES
Pentazocine
Content Navigation
CAS Number
Product Name
IUPAC Name
Molecular Formula
Molecular Weight
InChI
InChI Key
SMILES
solubility
1 G IN GREATER THAN 1000 ML WATER, 11 ML ALCOHOL, 2 ML CHLOROFORM, 42 ML ETHER
SOL IN ACETONE; SPARINGLY SOL IN ETHYL ACETATE
1.22e-01 g/L
Synonyms
Canonical SMILES
Isomeric SMILES
Analgesic Effects and Mechanism of Action
Research explores pentazocine’s interaction with different receptors in the nervous system to produce pain relief. Studies in rats suggest it works through kappa-opioid receptors and might have additional interactions with other neurotransmitter systems like acetylcholine ().
Comparison with Other Pain Medications
Studies compare pentazocine’s effectiveness with other pain medications. For example, a clinical trial investigated the use of rectal diclofenac combined with intramuscular pentazocine compared to just pentazocine for post-cesarean section pain relief ().
Potential for Abuse and Dependence
Research also focuses on the potential for abuse and dependence associated with pentazocine. Studies among people who inject drugs and patients with sickle cell disease highlight this concern, pointing to the need for stricter regulations and improved support systems to prevent misuse (, ).
Pentazocine is a synthetic opioid analgesic primarily used for the treatment of moderate to severe pain. It is classified as a partial agonist at the mu-opioid receptor and a full agonist at the kappa-opioid receptor. This dual action allows it to provide analgesic effects while also limiting some of the side effects commonly associated with full agonists like morphine. Pentazocine was first synthesized in 1958 and received approval from the Food and Drug Administration in 1967. It is marketed under various brand names, including Talwin, and is available in oral, intravenous, and intramuscular forms .
Key Reactions:
- Hydrolysis: Pentazocine can break down in acidic environments, leading to inactive metabolites.
- Decomposition: Under specific conditions (e.g., heat), pentazocine may form various degradation products.
Pentazocine exhibits significant biological activity through its interaction with opioid receptors. It primarily activates kappa-opioid receptors while having partial agonist activity at mu-opioid receptors. This unique profile contributes to its analgesic properties but also results in side effects such as hallucinations and dysphoria at higher doses . The onset of pain relief typically occurs within 15 to 30 minutes after administration, with effects lasting for several hours .
Pharmacokinetics:
The synthesis of pentazocine involves several steps that typically include the reaction of appropriate precursors under controlled conditions. The original synthesis pathway developed by Sterling Drug Company involved complex organic reactions that led to the formation of the pentazocine structure .
General Synthesis Overview:
- Starting Materials: Typically involves cyclic compounds that can undergo nucleophilic substitution.
- Reagents and Conditions: Specific reagents are used to facilitate the formation of the key bonds in the pentazocine structure.
- Purification: The final product is purified through crystallization or chromatography.
Pentazocine is primarily used as an analgesic for managing pain. Its unique receptor activity makes it suitable for patients who may be at risk for opioid dependence or those who experience adverse effects from traditional opioids. Additionally, it has been explored for use in treating symptoms of mania in bipolar disorder due to its action on dopamine pathways .
Clinical Uses:
- Pain management (moderate to severe).
- Potential off-label use in psychiatric disorders.
Pentazocine interacts with various other substances, which can enhance or diminish its effects. Notable interactions include:
- CNS Depressants: Co-administration with benzodiazepines or alcohol can increase the risk of respiratory depression and sedation.
- Opioid Antagonists: The presence of naloxone in certain formulations aims to deter misuse by blocking the euphoric effects if injected .
Adverse Effects:
Common side effects include nausea, dizziness, respiratory depression, and potential psychotomimetic effects such as hallucinations .
Pentazocine shares structural and functional similarities with several other opioid analgesics. Here are some notable compounds:
| Compound | Type | Unique Features |
|---|---|---|
| Butorphanol | Opioid Agonist/Antagonist | Stronger kappa receptor agonist; used as a nasal spray |
| Nalbuphine | Opioid Agonist/Antagonist | Less likely to cause euphoria; used for pain relief |
| Buprenorphine | Partial Agonist | Long duration of action; used in addiction treatment |
Uniqueness of Pentazocine:
While similar compounds like butorphanol and nalbuphine also act on kappa-opioid receptors, pentazocine’s unique profile as a partial mu-opioid receptor agonist differentiates it from others. Its propensity to cause psychotomimetic effects at higher doses further sets it apart from traditional opioids, which generally do not exhibit such properties
-
Buy Pentazocine | 359-83-1 | >98% – smolecule.com
Research explores pentazocine’s interaction with different receptors in the nervous system to produce pain relief.
-
Pentazocine 1.0mg/mL methanol, ampule 1mL, certified reference …
Pentazocine solution 1.0 mg/mL in methanol, ampule of 1 mL, certified reference material, Cerilliant®; CAS Number: 359-83-1; EC Number: 200-659-6 at Sigma-Aldrich
-
Brand: CERILLIAN
-
-
[Pentazocine CIV (500 mg)] – CAS [359-83-1] – USP
View the USP Certificate or Product Information Sheet in the table above to view additional product details, including available label text and storage information. 1 The Harmonized System (HS) code …
-
Buy Online CAS Number 359-83-1 – TRC – Pentazocine (1mg/ml in …
Purchase online CAS Number 359-83-1 – TRC – Pentazocine (1mg/ml in Methanol). High Quality CRMs, Reference Materials, Proficiency Testing & More at LGC Standards.
-
Buy Pentazocine (EVT-278515) | 359-83-1 – evitachem.com
Pentazocine is a synthetic opioid with both agonist and antagonist activity against opiate receptors which is used in oral and parenteral forms as an analgesic for moderate-to-severe pain.
-
PENTAZOCINE | 359-83-1 – ChemicalBook
Jan 13, 2026 · PENTAZOCINE (CAS 359-83-1) information, including chemical properties, structure, melting point, boiling point, density, formula, molecular …
-
FP26819 | 359-83-1 | Pentazocine | Biosynth
Buy Pentazocine FP26819 359-83-1 online for pharmaceutical testing. High-quality reference standards for accurate results.
-
(±)-Pentazocine (cis- (±)-Pentazocine, DL-Pentazocine, WIN 20,228, …
Formulations containing (±)-pentazocine have been used in the treatment of severe pain and as supplements to surgical anesthesia. (±)-Pentazocine is regulated as a Schedule IV compound in the …
-
Pentazocine | CAS 359-83-1 | Chemical-Suppliers
Pentazocine | CAS 359-83-1 REF CSP122324845411 – structural formula, chemical names, physical and chemical properties, references, safety/hazards/toxicity information, supplier lists, and more.
-
Pentazocine: CAS # 359-83-1 Compound Information and …



Reviews
There are no reviews yet.