Buy MBDB (entactogen) Cas 135795-90-3
Buy MBDB (entactogen) Cas 135795-90-3
MBDB, also known as N-methyl-1,3-benzodioxolylbutanamine or as 3,4-methylenedioxy-N-methyl-α-ethylphenylethylamine, is an entactogen of the phenethylamine, amphetamine, and phenylisobutylamine families related to MDMA.[1][2][3][4][5] It is known by the nicknames “Eden” and “Methyl-J“.[5]
Use and effects
MBDB was first synthesized by pharmacologist and medicinal chemist David E. Nichols and later tested by Alexander Shulgin and described in his book, PiHKAL: A Chemical Love Story.[1] MBDB’s dose, according to PiHKAL, is 180 to 210 mg; the proper dosage relative to body mass seems unknown.[1][2] Its duration is 4 to 8 hours, with noticeable after-effects lasting for 1 to 3 hours.[1][2]
MBDB was initially developed as a non-psychedelic entactogen. It has lower effects on the dopamine system in comparison to other entactogens such as MDMA. MBDB causes many mild, MDMA-like effects, in particular the lowering of social barriers and inhibitions, pronounced sense of empathy and compassion, and mild euphoria, all of which are present.[1] MBDB tends to produce less euphoria, psychedelia, and stimulation in comparison to MDMA.[1]
Clinical studies have found that MBDB produces similar entactogenic effects to MDMA, but lacks psychedelic and stimulant effects.[2][3] It enhances mood similarly to MDMA, but lacks the pronounced euphoria of MDMA.[2] MBDB produces prosocial effects similarly to MDMA, although it is said to be moderately less effective.
Buy MBDB (entactogen) Cas 135795-90-3
Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3,Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3
Interactions
Pharmacology
Pharmacodynamics
MBDB acts as a serotonin–norepinephrine releasing agent (SNRA).[2][3][6] Its EC50Tooltip half-maximal effective concentration values for induction of monoamine release are 540 nM for serotonin, 3,300 nM for norepinephrine (6.1-fold lower than for serotonin), and >100,000 for dopamine (>185-fold lower than for serotonin).[6] However, it may still have slight dopamine-releasing actions.[3] MBDB fully substitutes for MDMA in drug discrimination tests in rodents.[2][3] It increases locomotor activity similarly to but less robustly than MDMA.[7] Likewise, MBDB increases conditioned place preference (CPP) similarly but less efficaciously than MDMA.[3] In contrast to MDMA, which produced hyperthermia, MBDB instead produced dose- and time-dependent hypothermia.[7]
MBDB has similar affinities for the serotonin 5-HT1A and 5-HT2A receptors as MDMA.[2][8] However, MBDB did not show the head-twitch response, a behavioral proxy of psychedelic effects, at any dose in rodents.[7] In addition, MBDB (as well as MDMA) do not substitute for lysergic acid diethylamide (LSD) in drug discrimination tests.[3] The lack of apparent hallucinogenic effects with MBDB is analogous to the case of Ariadne, the α-ethyl homologue of DOM; (R)-Ariadne (BL-3912A) showed no psychedelic effects in humans at doses of up to 270 mg orally, whereas DOM is active as a hallucinogen at doses of 5 to 10 mg orally.[8][9] This may be due to lower activational efficacy at the serotonin 5-HT2A receptor.[9]
MBDB is a serotonergic neurotoxin similarly to MDMA.[2][3][10] However, MBDB appears to have reduced serotonergic neurotoxicity compared to MDMA at behaviorally equivalent doses.[2][3][10][4] In addition, unlike MDMA, MBDB does not produce dopaminergic neurotoxicity in mice.[10]
MBDB and its individual enantiomers, (S)-MBDB and (R)-MBDB, show similar behavioral effects in animals.[7]
Pharmacokinetics
The metabolism of MBDB has been described in the scientific literature.[11]
Chemistry
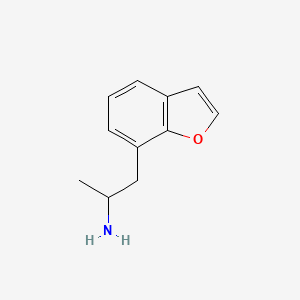
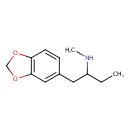
MBDB is a ring substituted amphetamine and an analogue of MDMA. Like MDMA, it has a methylenedioxy substitution at the 3 and 4 position on the aromatic ring; this is perhaps the most distinctive feature that structurally define analogues of MDMA, in addition to their unique effects, and as a class they are often referred to as “entactogens” to differentiate between typical stimulant amphetamines that (as a general rule) are not ring substituted.[citation needed] MBDB differs from MDMA by having an ethyl group instead of a methyl group attached to the alpha carbon. Modification at the alpha carbon is uncommon for substituted amphetamines.[citation needed]
Analogues
Analogues of MBDB (methyl-J) include BDB (J), EBDB (ethyl-J), BDP (K), MBDP (methyl-K), EBDP (ethyl-K), MPAP (PBDP; propyl-K), and UWA-101 (α-cyclopropyl-MDPEA), among others.[1]
History
MBDB was first described in the scientific literature by a group that included David E. Nichols, Robert A. Oberlender, Peyton Jacob III, and Alexander Shulgin in 1986.[12][13]
Society and culture
Legal status
International
Unlike MDMA, MBDB is not internationally scheduled under the United Nations Convention on Psychotropic Substances. The thirty-second meeting of the WHO Expert Committee on Drug Dependence (September 2000) evaluated MBDB and recommended against scheduling.[14] From the WHO Expert Committee assessment of MBDB:
- Although MBDB is both structurally and pharmacologically similar to MDMA, the limited available data indicate that its stimulant and euphoriant effects are less pronounced than those of MDMA. There have been no reports of adverse or toxic effects of MBDB in humans. Law enforcement data on illicit trafficking of MBDB in Europe suggest that its availability and abuse may now be declining after reaching a peak during the latter half of the 1990s. For these reasons, the Committee did not consider the abuse liability of MBDB would constitute a significant risk to public health, thereby warranting its placement under international control. Scheduling of MBDB was therefore not recommended.
-
N-methyl-1-(1,3-benzodioxol-5-yl)-2-butanamine Class Small Molecule Description 1,3-Benzodioxolyl-N-methylbutanamine (N-methyl-1,3-benzodioxolylbutanamine, MBDB, 3,4-methylenedioxy-N-methyl-α-ethylphenylethylamine) is an entactogen of the phenethylamine chemical class. It is known by the street names Eden and Methyl-J. MBDB is a closely related chemical analogue of MDMA, with the only difference between the two molecules being an ethyl group instead of a methyl group attached to the alpha carbon. It has IC50 values of 784 nM against 5-HT, 7825 nM against dopamine, and 1233 nM against norepinephrine. Its metabolism has been described in scientific literature.MBDB was first synthesized by pharmacologist and medicinal chemist David E. Nichols and later tested by Alexander Shulgin and described in his book, PiHKAL: A Chemical Love Story. MBDB’s dosage, according to PiHKAL, is 180–210 mg; the proper dosage relative to body mass seems unknown. Its duration is 4–6 hours, with noticeable after-effects lasting for 1–3 hours. MBDB was initially developed as a non-psychedelic entactogen. It has lower effects on the dopamine system in comparison to other entactogens such as MDMA. MBDB causes many mild, MDMA-like effects, in particular the lowering of social barriers and inhibitions, pronounced sense of empathy and compassion, mood lift, and mild euphoria, all of which are present. MBDB tends to produce less euphoria, psychedelia, and stimulation in comparison to MDMA. Contaminant Sources - STOFF IDENT Compounds
Contaminant Type Buy MBDB (entactogen) Cas 135795-90-3 Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3,Buy MBDB (entactogen) Cas 135795-90-3, Buy MBDB (entactogen) Cas 135795-90-3
Chemical Structure Synonyms Value Source N-Methyl-1-(1,3-benzodioxol-5-yl)-2-butanamine, (S)-isomer MeSH N-Methyl-1-(1,3-benzodioxol-5-yl)-2-butanamine MeSH N-Methyl-1-(1,3-benzodioxol-5-yl)-2-butanamine, (R)-isomer MeSH Chemical Formula C12H17NO2 Average Molecular Mass 207.273 g/mol Monoisotopic Mass 207.126 g/mol CAS Registry Number 135795-90-3 IUPAC Name [1-(2H-1,3-benzodioxol-5-yl)butan-2-yl](methyl)amine Traditional Name methylbenzodioxolylbutanamine SMILES CCC(CC1=CC2=C(OCO2)C=C1)NC InChI Identifier InChI=1S/C12H17NO2/c1-3-10(13-2)6-9-4-5-11-12(7-9)15-8-14-11/h4-5,7,10,13H,3,6,8H2,1-2H3 InChI Key USWVWJSAJAEEHQ-UHFFFAOYSA-N Chemical Taxonomy Description belongs to the class of organic compounds known as benzodioxoles. These are organic compounds containing a benzene ring fused to either isomers of dioxole. Dioxole is a five-membered unsaturated ring of two oxygen atoms and three carbon atoms. Kingdom Organic compounds Super Class Organoheterocyclic compounds Class Benzodioxoles Sub Class Not Available Direct Parent Benzodioxoles Alternative Parents Substituents - Benzodioxole
- Aralkylamine
- Benzenoid
- Oxacycle
- Secondary amine
- Secondary aliphatic amine
- Acetal
- Organic nitrogen compound
- Organic oxygen compound
- Organopnictogen compound
- Hydrocarbon derivative
- Organooxygen compound
- Organonitrogen compound
- Amine
- Aromatic heteropolycyclic compound
Molecular Framework Aromatic heteropolycyclic compounds
-
135795-90-3|DL-MBDB:HCL|Taizhou Zhenyu Biotech Co.,LTD
You can buy high quality, low price 135795-90-3 (DL-MBDB:HCL) here. We also provide a 135795-90-3 (DL-MBDB:HCL) database free of charge, including basic information, chemical properties, uses, …
-
CAS No.135795-90-3,DL-MBDB:HCL Suppliers – LookChem
Find quality suppliers and manufacturers of 135795-90-3 (DL-MBDB:HCL) for price inquiry. where to buy 135795-90-3 (DL-MBDB:HCL).Also offer free database of 135795-90-3 (DL-MBDB:HCL) including …
-
Buy DL-MBDB:HCL Industrial Grade,Pharma Grade from Hangzhou …
Hangzhou zhongqi chem co.,Ltd, with approximately 24443 visits to its shop, is a reputable Trader that has been providing DL-MBDB:HCL products for 2 years on ECHEMI.
-
DL-MBDB:HCL | 135795-90-3 – ChemicalBook
Visit ChemicalBook To find more DL-MBDB:HCL (135795-90-3) information like chemical properties,Structure,melting point,boiling point,density,molecular formula,molecular weight, physical …
-
1,3-Benzodioxole-5-ethanamine, α-ethyl-N-methyl- – ChemBK
Name:DL-MBDB:HCL,CAS:135795-90-3.Molecular Fomula:C12H17NO2,Molar Mass:207.27,Density:1.078±0.06 g/cm3 (Predicted),Boling Point:299.8±9.0 °C …
-
MBDB (CAS No. 135795-90-3) Suppliers – chemicalregister.com
Suppliers List, E-mail/RFQ Form, Molecular Structure, Weight, Formula, IUPAC, Synonyms for MBDB (CAS No. 135795-90-3)
-
ContaminantDB: N-methyl-1- (1,3-benzodioxol-5-yl)-2-butanamine
Your source for quantitative metabolomics technologies and bioinformatics. N-methyl-1- (1,3-benzodioxol-5-yl)-2-butanamine (CHEM043751)
-
MBDB – Chemical Route
The brain-based entactogen, methylbutyric dibuterol, was initially developed as a psychiatric entactogen. It has milder effects than other psychiatric entactogens, …
-
1,3-Benzodioxolyl-N-methylbutanamine | C12H17NO2 – ChemSpider
ChemSpider record containing structure, synonyms, properties, vendors and database links for 1,3-Benzodioxolyl-N-methylbutanamine, 135795-90-3, MBDB, USWVWJSAJAEEHQ-UHFFFAOYSA-N
-
DL-MBDB:HCL, CasNo.135795-90-3 HEBEI SHENGSUAN …
Inner double plastic bags—-5kg/Aluminum foil bag (GW: 6.5kg, NW: 5kg).3. Inner double plastic bags—-1kg/Aluminum foil bag (GW: 1.5kg, NW: 1kg).P.S.: we accept packaging customization



Reviews
There are no reviews yet.