4-Fluoroamphetamine (4-FA) Cas 459-02-9
4-Fluoroamphetamine (4-FA) Cas 459-02-9
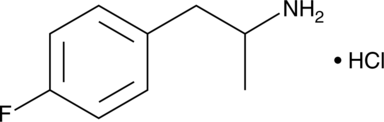
4-Fluoroamphetamine (4-FA; 4-FMP; PAL-303; “Flux“), also known as para-fluoroamphetamine (PFA), is a psychoactive research chemical of the phenethylamine and substituted amphetamine chemical classes. It produces stimulant and entactogenic effects. As a recreational drug, 4-FA is sometimes sold along with related compounds such as 2-fluoroamphetamine and 4-fluoromethamphetamine.
Key points about 4-FA:
-
Chemical Formula: C9H13ClFN
-
Molecular Weight: 189.66 g/mol
Usage

4-FA was popular in the Netherlands where it was predominantly used for its specific effects (77% of users) rather than its legal status (18%).[5] 4-FA has become illegal since May 2017.[6] Since the ban usage has decreased a lot. Accurate statistics of usage are not available until 2020, but submitted samples to get tested were counted. In 2016 1.100 (10%) of samples tested for DIMS were 4-FA samples,[7] in 2024 less than 50 4-FA samples were submitted.[8] In 2020 0,3% of Dutch adults used 4-FA in the previous year, in 2024 this was just 0,1%.[9]
Effects
The subjective effects of 4-fluoroamphetamine include euphoria which some find similar to the effects of MDMA and amphetamine,[5] increased energy (stimulation), mood elevation, feelings of warmth and empathy, excessive talking, bruxism, and suppressed appetite (anorexic). The general course of effects involves primarily empathogenic effects for the first few hours, which fades out as increased stimulation develops over the next several hours. [medical citation needed] 4-Fluoroamphetamine (4-FA) Cas 459-02-9
The dopamine reuptake inhibition produced by 4-FA is stronger than that of either 4-CA or 4-IA.[10] 4-FA also produces less hyperthermia than similar compounds such as PMA, 4-MTA and 4-methylamphetamine.[medical citation needed]
4-FA has been described as producing a very mild “psychedelic” state, intermediate between that of amphetamine and MDMA.[11] It is unclear whether this is related to induction of monoamine release or serotonin 5-HT2A receptor agonism.[11]
Common acute side effects are nausea, headaches, increased heart rate and insomnia.[medical citation needed]
Synonyms
- 4-
FA - p-FA
Technical Information
Regulatory Information
Shipping & Storage Information
Specification
The IUPAC name of 4-Fluoro-alpha-methylphenethylamine is 1-(4-fluorophenyl)propan-2-amine. With the CAS registry number 459-02-9, it is also named as Benzeneethanamine, p-fluoro-alpha-methyl- (9CI). The classification code is Drug / Therapeutic Agent. It is a psychoactive drug and research chemical of the phenethylamine and amphetamine chemical classes. Additioanlly, it was banned in Lithuania in July 2009. And it will be banned in Switzerland the 1st December 2010.
The other characteristics of 1-(4-Fluorophenyl)propane-2-amine can be summarized as:
(1)ACD/LogP: 1.86; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): -1.22; (4)ACD/LogD (pH 7.4): -0.53; (5)ACD/BCF (pH 5.5): 1; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 1; (8)ACD/KOC (pH 7.4): 1; (9)#H bond acceptors: 1; (10)#H bond donors: 2; (11)#Freely Rotating Bonds: 3; (12)Index of Refraction: 1.509; (13)Molar Refractivity: 43.92 cm3; (14)Molar Volume: 147 cm3; (15)Polarizability: 17.41×10-24 cm3; (16)Surface Tension: 34.8 dyne/cm; (17)Enthalpy of Vaporization: 45.15 kJ/mol; (18)Vapour Pressure: 0.15 mmHg at 25°C; (19)Rotatable Bond Count: 2; (20)Exact Mass: 153.095378; (21)MonoIsotopic Mass: 153.095378; (22)Topological Polar Surface Area: 26; (23)Heavy Atom Count: 11; (24)Complexity: 108.
Preparation of 1-(4-Fluorophenyl)propane-2-amine:
It can be obtained by 1-(4-fluorophenyl)-2-nitropropene. This reaction needs reagent LiAlH4 and solvent tetrahydrofuran at ambient temperature. The reaction time is 4.0 hours. The yield is 52%.
Uses of 1-(4-Fluorophenyl)propane-2-amine:
It can produce stimulant and possibly entactogenic effects. It is also a potent stimulant and serotonin releaser as with other para-substituted amphetamine derivatives, but is both significantly less potent as a serotonin releaser and much less neurotoxic than related compounds. In addition, this chemical can react with 3-bromo-propyne to get (R,S)-(+/-)-a-methyl-b-(4-fluorophenyl)-N-propynylethylamine. This reaction needs reagent K2CO3 and solvent acetonitrile, toluene and H2O. The yield is 62%.
People can use the following data to convert to the molecule structure.
1. SMILES:Fc1ccc(cc1)CC(N)C
2. InChI:InChI=1/C9H12FN/c1-7(11)6-8-2-4-9(10)5-3-8/h2-5,7H,6,11H2,1H3
3. InChIKey:DGXWNDGLEOIEGT-UHFFFAOYAT
The following are the toxicity data of 1-(4-Fluorophenyl)propane-2-amine:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intraperitoneal | 46mg/kg (46mg/kg) | “International Symposium on Amphetamines and Related Compounds, Proceedings, Mario Negri Institute for Pharmacological Research, Milan, 1969,” Costa, E., and S. Garattini, eds., New York, Raven Press, 1970Vol. -, Pg. 21, 1970. |
Chemistry
4-FA reacts with reagent testing to give a semi-unique array of colors which can be used to aid its identification.
| Reagent | Reaction color |
|---|---|
| Marquis | No reaction[12] |
| Mandelin | Pale Blue[12][13] |
| Liebermann | Orange[12][13] |
| Froehde | Faint purple/brown[12] or no reaction. |
Pharmacology
4-Fluoroamphetamine is a releasing agent and reuptake inhibitor of dopamine, serotonin, and norepinephrine.[14] The respective EC50 values are 200 nM, 730 nM, and 37 nM, while the IC50 values are 770 nM, 6800 nM, and 420 nM.[4]
The drug shows some affinity for the serotonin 5-HT2A receptor (Ki = 11,300 nM) and serotonin 5-HT2C receptor (Ki = 7,800 nM), similar to that of MDMA in the case of the serotonin 5-HT2A receptor (Ki = 5,900 nM), but far below the affinity of structurally related classical serotonergic psychedelics like 2C-B.[11][15] It has also been shown to act as a very low-potency serotonin 5-HT2B receptor partial agonist (EC50Tooltip half-maximal effective concentration = 14,400 nM; EmaxTooltip maximal efficacy = 58%).[15]
4-Fluoroamphetamine has been found to be a weak monoamine oxidase A (MAO-A) inhibitor, with an IC50Tooltip half-maximal inhibitory concentration of 16,000 nM.[16] For comparison, the IC50Tooltip half-maximal inhibitory concentration of amphetamine for MAO-A inhibition was 11,000 nM.[16]
Regarding the metabolic fate of 4-FA, the C-F bond at the 4-position on the phenyl ring likely resists deactivation in the liver by cytochrome P450 oxidase.[17][18]
Neurotoxicity
4-FA does not cause long-lasting depletion of brain serotonin, unlike its analogs 4-CA and 4-BA.[19] This is thought to “reflect the inability of the fluoro-compound to be metabolized in the same way as the other haloamphetamines.”[20]
Neurotoxicity does not increase down the series of para-halogenated amphetamine derivatives, even though serotonin releasing potency does follow this trend. For example, 4-iodoamphetamine is less toxic than is 4-chloroamphetamine.[10][21] Hence, this property is not related to serotonin releasing potency as such, since PAL-287 was reported to be not at all neurotoxic even though it is a powerful 5-HT releasing agent.[22] It is unclear where 4-methylamphetamine fits in on the neurotoxicity scale. The extensive serotonergic neurotoxicity of 4-chloroamphetamine (and its brominated derivative), and the increased serotonergic toxicity of 4-methylamphetamine[23] suggest that para-substitution seems to increase overall serotonergic (neuro)toxicity, compared to amphetamine. Exceptions include 4-MTA, a para-substituted, non-neurotoxic amphetamine.[24][25][26]
Toxicology
The LD50 (mouse; i.p.) of 4-FA is 46 mg/kg.[27]
Fluoroamphetamine (isomer not determined) in a capsule mixed with 25C-NBOMe was associated with three deaths in Melbourne in 2017.[28]
4-Fluoroamphetamine, para-fluoroamphetamine or 4-fluoro-alpha-methylphenethylamine (4-FMP), is a central nervous system stimulant. It is a chemical analog of amphetamine, and is also related to other phenethylamines, such as cathinone, methcathinone, and methamphetamine.
Effects of 4-FA
4-Fluoroamphetamine has a very short history of human use and is very uncommon so therefore little is known about its pharmacological effects, as is the case with many other designer drugs.
4-Fluoroamphetamine clearly has an amphetamine type of effect on the central nervous system. The halogenated compound also shows a mild serotogenic release in the user giving it a very slight resemblance to MDMA, it also differs from other halogenated amphetamines in that it is not known to be as neurotoxic compared with para-chloroamphetamine, para-bromoamphetamine and para-iodoamphetamine. It is noted that the potency of 4-FMP is roughly 2/3 that of dextroamphetamine. 4-FMP when insufflated tends to numb the nasal cavity and can be extremely painful, this way of administration should be avoided. It is suggested that this compound does not have the harsh come down like other CNS stimulants.
Effects include euphoria, increased energy, mood elevation, excessive talking, bruxism (jaw clenching) and suppressed appitite. It also inhibits the ability to sleep, like most stimulants. 4-Fluoroamphetamine (4-FA) Cas 459-02-9, 4-Fluoroamphetamine (4-FA) Cas 459-02-9, 4-Fluoroamphetamine (4-FA) Cas 459-02-9, 4-Fluoroamphetamine (4-FA) Cas 459-02-9, 4-Fluoroamphetamine (4-FA) Cas 459-02-9, 4-Fluoroamphetamine (4-FA) Cas 459-02-9, 4-Fluoroamphetamine (4-FA) Cas 459-02-9, 4-Fluoroamphetamine (4-FA) Cas 459-02-9, 4-Fluoroamphetamine (4-FA) Cas 459-02-9, 4-Fluoroamphetamine (4-FA) Cas 459-02-9, 4-Fluoroamphetamine (4-FA) Cas 459-02-9




Reviews
There are no reviews yet.