Buy 7-APB 7-(2-Aminopropyl) 286834-87-5
Buy 7-APB 7-(2-Aminopropyl) 286834-87-5
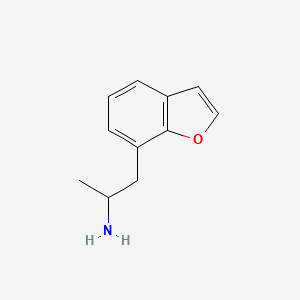
7-(2-Aminopropyl)benzofuran (7-APB) is a monoamine releasing agent and designer drug of the benzofuran family, albeit with a potency an order of magnitude less than 6-APB.[1][2] It acts as a serotonin–norepinephrine–dopamine releasing agent (SNDRA).[1][2] The drug was detected as a novel designer drug, in Europe, by 2013.
7-APB is a derivative of the designer drug 6-APB , also known as benzo fury, and is a stimulant and entactogen belonging to the amphetamine and the phenethylamine classes.[1] It is an analog of MDA where the 3,4-methylenedioxyphenyl ring system has been replaced with a benzofuran ring. This product is intended for forensic purposes.
Reference:
[1]. Stanczuk, A., Morris, N., Gardner, E.A., et al. Identification of (2-aminopropyl)benzofuran (APB) phenyl ring positional isomers in internet purchased products. Drug Test.Anal. 5(4), 270-276 (2013).
| Cas No. | 286834-86-4 | SDF | |
| Synonyms | 7-(2-Aminopropyl)Benzofuran | ||
| Chemical Name | α-methyl-7-benzofuranethanamine, monohydrochloride | ||
| Canonical SMILES | CC(N)CC1=CC=CC2=C1OC=C2.Cl | ||
| Formula | C11H13NO•HCl | M.Wt | 211.7 |
| Solubility | 20mg/mL in DMSO, 20mg/mL in DMF, 20mg/mL in Ethanol | Storage | Store at -20°C |
| General tips | Please select the appropriate solvent to prepare the stock solution according to the solubility of the product in different solvents; once the solution is prepared, please store it in separate packages to avoid product failure caused by repeated freezing and thawing.Storage method and period of the stock solution: When stored at -80°C, please use it within 6 months; when stored at -20°C, please use it within 1 month. To increase solubility, heat the tube to 37°C and then oscillate in an ultrasonic bath for some time. |
||
| Shipping Condition | Evaluation sample solution: shipped with blue ice. All other sizes available: with RT, or with Blue Ice upon request. | ||
| Prepare stock solution | |||
 |
1 mg | 5 mg | 10 mg |
| 1 mM | 4.7237 mL | 23.6183 mL | 47.2367 mL |
| 5 mM | 944.7 μL | 4.7237 mL | 9.4473 mL |
| 10 mM | 472.4 μL | 2.3618 mL | 4.7237 mL |
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
 g
g  μL
μL 
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Quality Control & SDS
- View current batch:
- Purity: >98.00%
- COA (Certificate Of Analysis)
- SDS (Safety Data Sheet)
- Datasheet
See also
-
Buy 7- (2-Aminopropyl)benzofuran | 286834-87-5
7- (2-Aminopropyl)benzofuran, also known as 7-APB, is a compound belonging to the class of substituted benzofurans. It has garnered attention due to its structural similarity to other psychoactive …
-
7-APB (hydrochloride) (7- (2-Aminopropyl)Benzofuran, CAS Number: …
7-APB (hydrochloride) (Item No. 14282) is an analytical reference standard categorized as a benzofuran. 7-APB is a potential metabolite of 7-MAPB (Item No. 14109). 1 At the time 7-APB (hydrochloride) …
-
7-Apb hydrochloride | C11H14ClNO | CID 10130547
7-Apb hydrochloride | C11H14ClNO | CID 10130547 – structure, chemical names, physical and chemical properties, classification, patents, literature, biological …
-
7-APB. 7- (2-Aminopropyl)Benzofuran is a derivative of 6-APB
7-APB is a derivative of the designer drug 6-APB , also known as benzo fury, and is a stimulant and entactogen belonging to the amphetamine and the phenethylamine classes.
-
7- (2-Aminopropyl)benzofuran | CAS#:286834-87-5 | Chemsrc
Chemsrc provides 7- (2-Aminopropyl)benzofuran (CAS#:286834-87-5) MSDS, density, melting point, boiling point, structure, formula, molecular weight etc. Articles of 7- (2-Aminopropyl)benzofuran are …
-
7-APB (hydrochloride) – 5 mg – Interprise
7-APB is a derivative of the designer drug 6-APB (Item No. 11079), also known as benzo fury, and is a stimulant and entactogen belonging to the amphetamine and the phenethylamine classes. {22885} It …
-
7-APB (hydrochloride) – Analytical Standards – CAT N°: 14282
7-APB (hydrochloride) to order online from our supplier Cayman, specialist in Analytical Standards. CAS Number: 286834-86-4. Size: 1 mg, 10 mg, 5 mg. Shipping: dry ice. UNSPSC Code: 12352100.
-
7-APB (hydrochloride) – Labchem Catalog
An analytical reference standard categorized as a benzofuran; a potential metabolite of 7-MAPB based on the published metabolism of 5-MAPB; intended …
-
7- (2-Aminopropyl)benzofuran (7-APB) is a monoamine releasing agent and designer drug of the benzofuran family, albeit with a potency an order of …
-
7-APB (hydrochloride) | Cas# 286834-86-4 – GlpBio7-APB is a derivative of the designer drug 6-APB, also known as benzo fury, and is a stimulant and entactogen belonging to the amphetamine and the phenethylamine classes.



Reviews
There are no reviews yet.