Buy Paroxetine (Paxil) Cas 61869-08-7
Buy Paroxetine (Paxil) Cas 61869-08-7
Paroxetine, sold under the brand name Paxil among others, is an antidepressant medication of the selective serotonin reuptake inhibitor (SSRI) class[6] used to treat major depressive disorder, obsessive–compulsive disorder (OCD), panic disorder, social anxiety disorder, post-traumatic stress disorder (PTSD), generalized anxiety disorder, and premenstrual dysphoric disorder.[6] It has also been used in the treatment of premature ejaculation, and hot flashes due to menopause.[6][7] It is taken orally (by mouth).[6]
Common side effects include drowsiness, dry mouth, loss of appetite, sweating, trouble sleeping, and sexual dysfunction.[6] Serious side effects may include suicidal thoughts in those under the age of 25, serotonin syndrome, and mania.[6] While the rate of side effects appears similar compared to other SSRIs and SNRIs, antidepressant discontinuation syndrome may occur more often.[8][9] Use in pregnancy is not recommended, while use during breastfeeding is relatively safe.[10] It is believed to work by blocking the reuptake of the chemical serotonin by neurons in the brain.[6]
Paroxetine was approved for medical use in the United States in 1992 and initially sold by GlaxoSmithKline.[6][11] It is on the World Health Organization’s List of Essential Medicines.[12] It is available as a generic medication.[13] In 2023, it was the 72nd most commonly prescribed medication in the United States, with more than 9 million prescriptions.[14][15] In 2018, it was in the top 10 of most prescribed antidepressants in the United States.[16]
Medical uses
Paroxetine is primarily used to treat major depressive disorder, obsessive–compulsive disorder, post-traumatic stress disorder, social anxiety disorder, and panic disorder. It is also occasionally used for agoraphobia, generalized anxiety disorder, premenstrual dysphoric disorder, and menopausal hot flashes.[17][18][19][20][21]
Depression
A variety of meta-analyses have been conducted to evaluate the efficacy of paroxetine in depression. They have variously concluded that paroxetine is superior to placebo and that it is equivalent to other antidepressants.[22][23][24] Despite this, there was no clear evidence that paroxetine was better or worse compared with other antidepressants at increasing response to treatment at any point in time. Buy Paroxetine (Paxil) Cas 61869-08-7
Anxiety disorders
Paroxetine was the first antidepressant approved in the United States for the treatment of panic disorder.[26][page needed] Several studies have concluded that paroxetine is superior to placebo in the treatment of panic disorder.[24][27]
Paroxetine has demonstrated efficacy for the treatment of social anxiety disorder in adults and children.[28][29] It is also beneficial for people with co-occurring social anxiety disorder and alcohol use disorder.[30] It appears to be similar to a number of other SSRIs.[31]
Paroxetine is used in the treatment of obsessive-compulsive disorder.[32] Comparative efficacy of paroxetine is equivalent to that of clomipramine and venlafaxine.[33][34] Paroxetine is also effective for children with obsessive-compulsive disorder.[35]
Paroxetine is approved for the treatment of PTSD in the United States, Japan, and Europe.[36][37][38] In the United States, it is approved for short-term use.[37]
Paroxetine is also FDA-approved for generalized anxiety disorder.[39]
Menopausal hot flashes
In 2013, low-dose paroxetine was approved in the US for the treatment of moderate-to-severe vasomotor symptoms such as hot flashes and night sweats associated with menopause.[7] At the low dose used for menopausal hot flashes, side effects are similar to placebo and dose tapering is not required for discontinuation.[40]
Fibromyalgia
Studies have also shown paroxetine “appears to be well-tolerated and improve the overall symptomatology in patients with fibromyalgia”, but is less robust in helping with the pain involved.[41][42]
Adverse effects
Common side effects include drowsiness, dry mouth, loss of appetite, sweating, insomnia, and sexual dysfunction.[6] Serious side effects may include suicide in those under the age of 25, serotonin syndrome, and mania.[6] While the rate of side effects appears similar compared to other SSRIs and SNRIs, antidepressant discontinuation syndromes may occur more often.[8][9] Use in pregnancy is not recommended, while use during breastfeeding is relatively safe.[10]
The Federal Aviation Administration (FAA), the U.S. agency responsible for regulating civil aviation, considers paroxetine to be an antidepressant medication that is ineligible for an FAA Authorization of Special Issuance (SI) or Special Consideration (SC) of a medical certificate.[43]
Paroxetine shares many of the common adverse effects of SSRIs, including (with the corresponding rates seen in people treated with placebo in parentheses):
- nausea 26% (9%)
- somnolence 23% (9%)
- dry mouth 18% (12%)
- headache 18% (17%)
- asthenia (weakness) 15% (6%)
- constipation 14% (9%)
- dizziness 13% (6%)
- insomnia 13% (6%)
- diarrhea 12% (8%)
- sweating 11% (2%)
- tremor 8% (2%)
- loss of appetite 6% (2%)
- nervousness 5% (3%)
- blurred vision 4% (1%)
- paraesthesia 4% (2%)
- hypomania 1% (0.3%)
- sexual dysfunction (≥10% incidence).[5]
Most of these adverse effects are transient and go away with continued treatment. Central and peripheral 5-HT3 receptor stimulation is believed to result in the gastrointestinal effects observed with SSRI treatment.[44] Compared to other SSRIs, it has a lower incidence of diarrhea, but a higher incidence of anticholinergic effects (e.g., dry mouth, constipation, blurred vision, etc.), sedation/somnolence/drowsiness, sexual side effects, and weight gain.[45]
Due to reports of adverse withdrawal reactions
-
To buy Paroxetine (Paxil) with CAS number 61869-08-7, you can consider the following options:
Paroxetine is a selective serotonin reuptake inhibitor (SSRI) commonly prescribed for the treatment of depression, anxiety disorders, and obsessive-compulsive disorder (OCD). By increasing serotonin levels in the brain, it helps improve mood, alleviate anxiety, and reduce obsessive behaviors. Paroxetine is also used to manage symptoms of post-traumatic stress disorder (PTSD) and premenstrual dysphoric disorder (PMDD). Its efficacy and well-established safety profile make it a widely used antidepressant, contributing to the mental health management of millions worldwide.
CAS Number 61869-08-7 Synonyms Paxil; Paxil CR; Casbol. Molecular Formula C19H20FNO3 Purity ≥95% Target GPCR/G Protein IUPAC Name (3S,4R)-3-(1,3-benzodioxol-5-yloxymethyl)-4-(4-fluorophenyl)piperidine InChI InChI=1S/C19H20FNO3/c20-15-3-1-13(2-4-15)17-7-8-21-10-14(17)11-22-16-5-6-18-19(9-16)24-12-23-18/h1-6,9,14,17,21H,7-8,10-12H2/t14-,17-/m0/s1 InChIKey AHOUBRCZNHFOSL-YOEHRIQHSA-N SMILES C1CNCC(C1C2=CC=C(C=C2)F)COC3=CC4=C(C=C3)OCO4 Chemistry Calculators Dilution Calculator
In vivo Formulation Calculator
Molarity Calculator
Molecular Weight Calculator
Reconstitution CalculatorParoxetine | S538652 | >98% (or refer to the COA) | smoleculeParoxetine
Catalog No.S538652CAS No.61869-08-7M.FC19H20FNO3M. Wt329.4 g/molAvailabilityIn Stock* This item is exclusively intended for research purposes and is not designed for human therapeutic applications or veterinary use.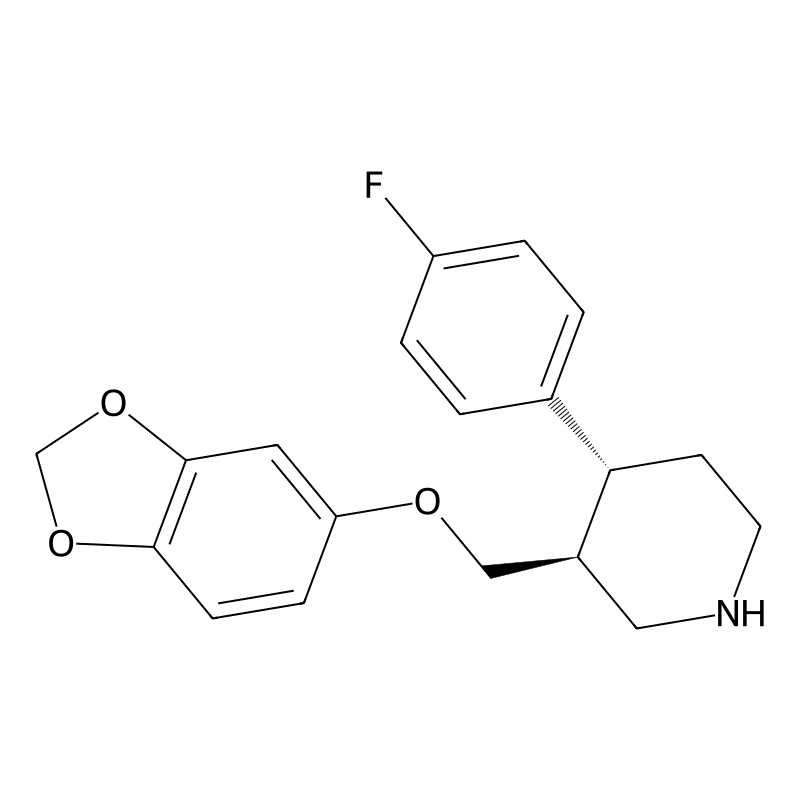
Content Navigation
CAS Number
61869-08-7Product Name
ParoxetineIUPAC Name
(3S,4R)-3-(1,3-benzodioxol-5-yloxymethyl)-4-(4-fluorophenyl)piperidineMolecular Formula
C19H20FNO3Molecular Weight
329.4 g/molInChI
InChI=1S/C19H20FNO3/c20-15-3-1-13(2-4-15)17-7-8-21-10-14(17)11-22-16-5-6-18-19(9-16)24-12-23-18/h1-6,9,14,17,21H,7-8,10-12H2/t14-,17-/m0/s1InChI Key
AHOUBRCZNHFOSL-YOEHRIQHSA-NSMILES
C1CNCC(C1C2=CC=C(C=C2)F)COC3=CC4=C(C=C3)OCO4solubility
Odorless, off-white powder, mp 147-150 °C . Solubility in water: >1 g/mL/Paroxetine methanesulfonate/
In water, 1,131 mg/L at 25 °C
8.53e-03 g/LSynonyms
Aropax, BRL 29060, BRL-29060, BRL29060, FG 7051, FG-7051, FG7051, Paroxetine, Paroxetine Acetate, Paroxetine Hydrochloride, Paroxetine Hydrochloride Anhydrous, Paroxetine Hydrochloride Hemihydrate, Paroxetine Hydrochloride, Hemihydrate, Paroxetine Maleate, Paroxetine, cis-(+)-Isomer, Paroxetine, cis-(-)-Isomer, Paroxetine, trans-(+)-Isomer, Paxil, SeroxatCanonical SMILES
C1CNCC(C1C2=CC=C(C=C2)F)COC3=CC4=C(C=C3)OCO4Isomeric SMILES
C1CNC[C@H]([C@@H]1C2=CC=C(C=C2)F)COC3=CC4=C(C=C3)OCO4The exact mass of the compound Paroxetine is 329.1427 and the complexity rating of the compound is unknown. The solubility of this chemical has been described as 5.4 mg/mlin water, 1,131 mg/l at 25 °c8.53e-03 g/l. Its Medical Subject Headings (MeSH) category is Diseases Category – Chemically-Induced Disorders – Drug-Related Side Effects and Adverse Reactions – Metabolic Side Effects of Drugs and Substances – Cytochrome P-450 Enzyme Inhibitors – Cytochrome P-450 CYP2D6 Inhibitors. It belongs to the ontological category of piperidines in the ChEBI Ontology tree. The United Nations designated GHS hazard class pictogram is Irritant;Health Hazard;Environmental Hazard, and the GHS signal word is DangerThe storage condition is described as Dry, dark and at 0 – 4 C for short term (days to weeks) or -20 C for long term (months to years).. Use and application categories indicated by third-party sources: Human Drugs -> FDA Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book) -> Active Ingredients. However, this does not mean our product can be used or applied in the same or a similar way.Understanding Mood Disorders:
Paroxetine’s mechanism of action, which involves increasing serotonin levels in the brain, has been instrumental in researching the neurochemical basis of mood disorders like depression and anxiety. Studies employing Paroxetine investigate how altering serotonin function impacts symptoms and explore potential new treatment targets [].
Exploring Anxiety Disorders:
Paroxetine’s effectiveness in treating various anxiety disorders, including generalized anxiety disorder, panic disorder, and social anxiety disorder, makes it a valuable tool in related research. Studies use Paroxetine to understand the neural mechanisms underlying these conditions and evaluate the efficacy of new treatment approaches [, ].
Investigating Obsessive-Compulsive Disorder (OCD):
Paroxetine is also approved for treating OCD, a condition characterized by intrusive thoughts and repetitive behaviors. Research utilizes Paroxetine to examine the role of serotonin in OCD pathophysiology and develop improved treatment strategies for this complex disorder [].
Paroxetine is a potent antidepressant belonging to the class of selective serotonin reuptake inhibitors (SSRIs). It is primarily used to treat major depressive disorder, anxiety disorders, obsessive-compulsive disorder, and post-traumatic stress disorder. The chemical structure of paroxetine is characterized by its unique piperidine ring and a benzodioxole moiety, which contribute to its pharmacological properties. The molecular formula for paroxetine is C19H20FNO3, with a molecular weight of approximately 329.3654 g/mol .
Paroxetine’s mechanism of action revolves around its interaction with the serotonin transporter protein. This protein is responsible for reabsorbing serotonin from the synaptic cleft, a space between neurons where neurotransmitters like serotonin transmit signals. By binding to the serotonin transporter, paroxetine prevents it from reuptaking serotonin, leading to increased levels of serotonin in the synapse []. This enhanced serotonin availability is believed to contribute to the antidepressant and anxiolytic effects of paroxetine [].
Paroxetine is generally well-tolerated, but common side effects include nausea, dizziness, fatigue, and sexual dysfunction []. In rare cases, it can cause suicidal ideation, particularly in younger patients []. Paroxetine can also interact with other medications, so it’s crucial to disclose all medications to a doctor before starting treatment [].
, primarily involving its metabolism in the liver. The drug is extensively metabolized by cytochrome P450 enzymes, particularly CYP2D6, leading to the formation of several metabolites. These metabolites include catechol derivatives that are further conjugated to form glucuronide and sulfate metabolites, which are significantly less potent than the parent compound .The primary reaction pathways for paroxetine include:
Paroxetine exhibits its biological activity primarily through the inhibition of serotonin reuptake in the central nervous system. By blocking the serotonin transporter, paroxetine increases the availability of serotonin in synaptic clefts, enhancing mood and emotional regulation. It also interacts with other neurotransmitter systems, albeit to a lesser extent, including norepinephrine .
Notably, paroxetine has been shown to bind at various sites on the serotonin transporter with high affinity, stabilizing its conformation and inhibiting serotonin reuptake effectively . Additionally, paroxetine has demonstrated inhibitory effects on G protein-coupled receptor kinase 2 (GRK2), which may contribute to its therapeutic effects .
The synthesis of paroxetine involves several steps that typically include the formation of the piperidine ring and subsequent functionalization to introduce the benzodioxole group. One common synthetic route involves:
- Formation of the piperidine core: Starting from appropriate precursors such as substituted phenols.
- Introduction of the benzodioxole moiety: This can be achieved through coupling reactions or C–H functionalization strategies.
- Fluorination: The introduction of the fluorine atom on the phenyl ring is crucial for enhancing its pharmacological profile .
Recent studies have explored novel synthetic routes that involve transition metal-catalyzed reactions to improve yield and selectivity .
Paroxetine is primarily used in clinical settings for:
- Major Depressive Disorder: Effective in alleviating symptoms associated with depression.
- Anxiety Disorders: Used to manage generalized anxiety disorder and social anxiety disorder.
- Obsessive-Compulsive Disorder: Helps reduce compulsive behaviors and intrusive thoughts.
- Post-Traumatic Stress Disorder: Assists in managing symptoms related to traumatic experiences.
Additionally, paroxetine is sometimes prescribed off-label for conditions such as premenstrual dysphoric disorder and chronic pain syndromes .
Paroxetine exhibits significant interactions with various medications due to its effects on cytochrome P450 enzymes. Notable interactions include:
- CYP2D6 Inhibition: Paroxetine is a potent inhibitor of this enzyme, affecting the metabolism of other drugs processed by CYP2D6 .
- Serotonin Syndrome Risk: Concurrent use with other serotonergic agents (e.g., triptans, monoamine oxidase inhibitors) may increase the risk of serotonin syndrome .
- Anticoagulants: Increased bleeding risk when used with anticoagulants like warfarin due to altered metabolism .
Paroxetine shares structural and functional similarities with other SSRIs but has unique attributes that distinguish it from them. Here are some comparable compounds:
Compound Unique Features Sertraline More balanced reuptake inhibition across serotonin and norepinephrine; fewer withdrawal symptoms. Fluoxetine Longer half-life; less potent inhibition of serotonin reuptake compared to paroxetine. Escitalopram More selective for serotonin receptors; fewer side effects reported compared to paroxetine. Citalopram Similar efficacy but different side effect profiles; less potent than paroxetine in serotonin reuptake inhibition. Paroxetine’s unique pharmacokinetic properties include a rapid onset of action and a higher likelihood of withdrawal effects upon discontinuation compared to some other SSRIs . Its binding affinity and interaction patterns with the serotonin transporter also contribute to its distinct therapeutic profile .
Physical State and Appearance
Paroxetine hydrochloride exists as a solid crystalline material at room temperature. The compound presents as an odorless, off-white powder with a characteristic appearance [1]. The physical state remains stable under normal storage conditions, maintaining its crystalline structure when properly stored [2].
The compound exhibits polymorphism, existing in multiple crystal forms including the stable hemihydrate form (Form I) and the anhydrous form (Form II) [3]. The hemihydrate form is considered the more stable configuration under standard conditions and is the form typically encountered in pharmaceutical preparations [4].
Melting and Boiling Points
Paroxetine hydrochloride demonstrates a melting point range of 120°C to 138°C [1] [5]. This relatively broad melting range is characteristic of the hemihydrate form and reflects the presence of water molecules within the crystal lattice structure. The melting point can vary slightly depending on the specific crystal form and hydration state of the compound [6].
The boiling point of paroxetine has been calculated to be 451.7±45.0°C [4] [7]. This high boiling point is consistent with the compound’s molecular weight and intermolecular forces. The relatively high boiling point indicates strong intermolecular interactions and suggests limited volatility under normal conditions [8].
Solubility Profiles
Aqueous Solubility
Paroxetine hydrochloride exhibits limited aqueous solubility, with a reported solubility of 5.4 mg/mL in water [1] [5]. This solubility decreases significantly at physiological pH conditions, where the compound demonstrates a solubility of approximately 0.00853 mg/mL at pH 7.4 [4]. The reduced solubility at physiological pH is attributed to the ionization state of the compound, as paroxetine has a pKa value of 9.9 [9] [10].
The aqueous solubility is influenced by pH conditions, with better solubility observed under acidic conditions where the amine group becomes protonated [10]. At pH 3.0, the compound shows improved dissolution characteristics compared to neutral or basic pH conditions [10].
Organic Solvent Solubility
Paroxetine demonstrates significantly higher solubility in organic solvents compared to aqueous systems. The compound exhibits solubility of approximately 20 mg/mL in both ethanol and dimethyl sulfoxide (DMSO) [2] [3]. In dimethyl formamide (DMF), paroxetine shows the highest solubility among tested solvents at approximately 33 mg/mL [2] [3].
For pharmaceutical applications requiring aqueous formulations, paroxetine can be dissolved in DMF and then diluted with aqueous buffer systems. Using this approach, the compound achieves a solubility of approximately 0.09 mg/mL in a 1:10 DMF:PBS solution at pH 7.0 [2] [3].
Paroxetine is entirely soluble in methanol, making it an excellent solvent for analytical applications [9]. The compound is sparingly soluble in methylene chloride, indicating limited compatibility with chlorinated solvents [9].
Partition Coefficient and Lipophilicity
Paroxetine exhibits significant lipophilic character, as demonstrated by its partition coefficient values. The compound has a log P value of 2.53 [4] and a log Po/w value of 3.95 [9] [11]. These values indicate strong preference for lipophilic environments over aqueous phases, which is consistent with its ability to cross biological membranes effectively.
The high lipophilicity is attributed to the presence of both hydrophobic and hydrophilic moieties within the molecular structure [9]. The lipophilic character facilitates the compound’s distribution throughout the body, including penetration into the central nervous system [9] [12].
Spectroscopic Properties
Paroxetine exhibits characteristic spectroscopic properties that enable its identification and quantification. The compound shows ultraviolet absorption maxima at 235, 265, 271, and 295 nm [13]. These absorption bands are associated with the aromatic ring systems present in the molecular structure, particularly the benzodioxol and fluorophenyl moieties.
Infrared spectroscopy reveals characteristic absorption bands consistent with the functional groups present in paroxetine [9] [14]. The compound shows specific absorption patterns in the 600-3500 cm⁻¹ range, with notable peaks corresponding to C-H stretching, aromatic C=C stretching, and C-O stretching vibrations [14].
Nuclear magnetic resonance spectroscopy provides detailed structural information, with both ¹H NMR and ¹³C NMR spectra being well-documented for paroxetine [9] [15]. The NMR data confirms the presence of the piperidine ring, benzodioxol system, and fluorophenyl group within the molecular structure [15].
Dissociation Constants
Paroxetine exhibits a pKa value of 9.9 [9] [10], indicating that it behaves as a weak base under physiological conditions. This high pKa value means that paroxetine exists predominantly in its protonated form at physiological pH (7.4), which influences its solubility, membrane permeability, and pharmacokinetic properties [10].
The dissociation constant suggests that paroxetine is a non-protonated compound under most analytical conditions and falls beyond the typical working pH range of chromatographic columns [10]. This characteristic affects its behavior in analytical systems and pharmaceutical formulations [10].
Collision Cross Section Properties
Paroxetine demonstrates specific collision cross section values that are useful for mass spectrometric identification and analysis. The compound exhibits a collision cross section of 175.6 Ų for the [M+H]⁺ adduct [16]. Other adduct forms show varying collision cross section values: [M+Na]⁺ at 181.1 Ų, [M-H]⁻ at 182.6 Ų, [M+NH₄]⁺ at 186.9 Ų, and [M+K]⁺ at 177.8 Ų [16].
These collision cross section values are particularly valuable for ion mobility spectrometry-mass spectrometry applications and provide additional selectivity for analytical methods [16]. The variation in collision cross section values reflects the different conformational states and charge distributions associated with different adduct formation [16].
Purity
>98% (or refer to the Certificate of Analysis) ( Purity information is a general guideline based on past batches. Please refer to your quote for the exact purity of the batch. )Physical Description
SolidXLogP3
3.5Hydrogen Bond Acceptor Count
5Hydrogen Bond Donor Count
1Exact Mass
329.14272166 g/molMonoisotopic Mass
329.14272166 g/molBoiling Point
451.7±45.0Heavy Atom Count
24LogP
2.53
log Kow = 1.23
3.6Appearance
Solid powderMelting Point
120-138
Odorless off-white powder; molecular weight: 374.84; melting point: 120-138 °C /Hydrochloride hemihydrate/
129 – 131 °CStorage
Dry, dark and at 0 – 4 C for short term (days to weeks) or -20 C for long term (months to years).UNII
41VRH5220H
32Q7TW8BX7Related CAS
64006-44-6 (maleate)
78246-49-8 (hydrochloride)
110429-35-1 (HCl hemihydrate)
72471-80-8 (acetate)GHS Hazard Statements
Aggregated GHS information provided by 6 companies from 4 notifications to the ECHA C&L Inventory. Each notification may be associated with multiple companies.;
Reported as not meeting GHS hazard criteria by 1 of 6 companies. For more detailed information, please visit ECHA C&L website;
Of the 3 notification(s) provided by 5 of 6 companies with hazard statement code(s):;
H302 (80%): Harmful if swallowed [Warning Acute toxicity, oral];
H360 (60%): May damage fertility or the unborn child [Danger Reproductive toxicity];
H361 (20%): Suspected of damaging fertility or the unborn child [Warning Reproductive toxicity];
H400 (20%): Very toxic to aquatic life [Warning Hazardous to the aquatic environment, acute hazard];
H410 (60%): Very toxic to aquatic life with long lasting effects [Warning Hazardous to the aquatic environment, long-term hazard];
Information may vary between notifications depending on impurities, additives, and other factors. The percentage value in parenthesis indicates the notified classification ratio from companies that provide hazard codes. Only hazard codes with percentage values above 10% are shown.Use and Manufacturing
Paroxetine is used as a medication or supplement. An overview is available from MedlinePlus, a consumer health web site produced by the National Library of Medicine.Drug Indication
Paroxetine is indicated for the management of depression, obsessive-compulsive disorder, panic disorder, social anxiety disorder, generalized anxiety disorder, posttraumatic stress disorder. One form of paroxetine, commercially known as Brisdelle, is used to manage mild to moderate vasomotor symptoms of menopause. Off-label, paroxetine may be used for the treatment of premature ejaculation or irritable bowel syndrome (IBS).Livertox Summary
Paroxetine is a selective serotonin reuptake inhibitor (SSRI) used in the therapy of depression, anxiety disorders and obsessive-compulsive disorder. Paroxetine therapy can be associated with transient asymptomatic elevations in serum aminotransferase levels and has been linked to rare instances of clinically apparent acute liver injury.Drug Classes
Antidepressant AgentsTherapeutic Uses
Antidepressive Agents, Second-Generation; Serotonin Uptake Inhibitors
/CLINICAL TRIALS/ ClinicalTrials.gov is a registry and results database of publicly and privately supported clinical studies of human participants conducted around the world. The Web site is maintained by the National Library of Medicine (NLM) and the National Institutes of Health (NIH). Each ClinicalTrials.gov record presents summary information about a study protocol and includes the following: Disease or condition; Intervention (for example, the medical product, behavior, or procedure being studied); Title, description, and design of the study; Requirements for participation (eligibility criteria); Locations where the study is being conducted; Contact information for the study locations; and Links to relevant information on other health Web sites, such as NLM’s MedlinePlus for patient health information and PubMed for citations and abstracts for scholarly articles in the field of medicine. Paroxetine is included in the database.
Paxil is indicated for the treatment of major depressive disorder. /Included in US product labeling/
Paxil is indicated for the treatment of obsessions and compulsions in patients with obsessive compulsive disorder (OCD) as defined in the DSM-IV. The obsessions or compulsions cause marked distress, are time-consuming, or significantly interfere with social or occupational functioning. /Included in US product labeling/
For more Therapeutic Uses (Complete) data for PAROXETINE (13 total), please visit the HSDB record page.Pharmacology
Paroxetine treats the symptoms of depression, various anxiety disorders, posttraumatic stress disorder, obsessive-compulsive disorder, and the vasomotor symptoms of menopause via the inhibition of serotonin reuptake.[T653,L3358,L7703] The onset of action of paroxetine is reported to be approximately 6 weeks.[A181874] Due its serotonergic activity, paroxetine, like other SSRI drugs, may potentiate serotonin syndrome. This risk is especially high when monoamine oxidase (MAO) inhibitors are given within 2 weeks of paroxetine administration. Upon cessation of MAO inhibitors, a 2-week interval before paroxetine administration is recommended. Do not coadminister these agents.[T656]MeSH Pharmacological Classification
Serotonin Uptake InhibitorsATC Code
N – Nervous system
N06 – Psychoanaleptics
N06A – Antidepressants
N06AB – Selective serotonin reuptake inhibitors
N06AB05 – ParoxetineMechanism of Action
Paroxetine enhances serotonergic activity via the inhibition presynaptic reuptake of serotonin by the serotonin (SERT) receptor. This inhibition raises the level of serotonin in the synaptic cleft, relieving various symptoms. This drug has been demonstrated to be a stronger inhibitor of serotonin reuptake than other members of the same drug class, including [Citalopram], [Fluoxetine], and [Fluvoxamine]. The mechanism of action of paroxetine in relieving the vasomotor symptoms of menopause is unknown, according to the Brisdelle prescribing information, but may occur due to its effects on thermoregulation. Paroxetine shows a clinically insignificant affinity for adrenergic alpha-1 and alpha-2 receptors and β-adrenergic receptors, dopamine D1 and D2 receptors, histamine H1 receptors and serotonin 5-HT1A, 5-HT2A and 5-HT2C receptors. This drug shows some affinity for muscarinic cholinergic receptors and 5-H2B receptors. The delayed onset of paroxetine therapeutic effects may be explained by the initial paroxetine actions on the 5-HT neurons. In rats, paroxetine activates 5-HT1A receptors when it is first administered, inhibiting the stimulation of the 5-HT neurons and subsequent release of serotonin at the synaptic cleft.
Functional and structural approaches were used to examine the inhibitory mechanisms and binding site location for fluoxetine and paroxetine, two serotonin selective reuptake inhibitors, on nicotinic acetylcholine receptors (AChRs) in different conformational states. The results establish that: (a) fluoxetine and paroxetine inhibit h alpha1beta1 gammadelta AChR-induced Ca(2+) influx with higher potencies than dizocilpine. The potency of fluoxetine is increased approximately 10-fold after longer pre-incubation periods, which is in agreement with the enhancement of (3)H-cytisine binding to resting but activatable Torpedo AChRs elicited by these -
Paroxetine | 61869-08-7 – Chemicea
Buy high quality Paroxetine from Chemicea Pharmaceuticals. Chemicea Pharmaceuticals is one of the leading manufacturer and exporter of Paroxetine.
-
Paroxetine | S538652 | >98% (or refer to the COA) | smolecule
Buy Paroxetine (CAS No. 61869-08-7) from Smolecule. Purity: >98% (or refer to the Certificate of Analysis).Molecular Formula: C19H20FNO3.
-
Paroxetine CAS#: 61869-08-7 – ChemicalBook
Paroxetine is a new highly selective serotonin reuptake inhibitor, mechanistically similar to fluoxetine, fluvoxamine and sertraline, introduced for the treatment of all types of depressive illnesses including …
-
CAS: 61869-08-7
-
MW: 329.37
-
MF: C19H20FNO3
-
Product Name: Paroxetine
-
-
Paroxetine | CAS 61869-08-7 | SCBT – Santa Cruz Biotechnology
Paroxetine (CAS 61869-08-7), MW: 329.369, MF: C19H20FNO3. Paroxetine. Over 170,000 chemicals at Santa Cruz!
-
CAS No : 61869-08-7 | Product Name : Paroxetine
Paroxetine intermediate. The PASL product information for any accuracy or …
-
Reviews: 5
-
Catalogue number: PA 16 3060000
-
CAS Number: 61869-08-7
-
Chemical name: Paroxetine
-
-
Where to Buy CAS No.: 61869-08-7 | Paroxetine
Paroxetine is a selective serotonin reuptake inhibitor (SSRI) commonly prescribed for the treatment of depression, anxiety disorders, and obsessive-compulsive disorder (OCD). By increasing serotonin …
-
Paroxetine | CAS No- 61869-08-7 | GLP Pharma Standards
Buy high quality Paroxetine, 61869-08-7 from GLP Pharma Standards, a perfect choice for Pharmaceutical Impurities, API relate
BP164263 | 61869-08-7 | Paroxetine HCl hemihydrate – Bio-X – Biosynth
Buy Paroxetine HCl hemihydrate – Bio-X ™ BP164263 61869-08-7 online for pharmaceutical testing. High-quality reference standards for accurate results.
-
Paroxetine – 97% prefix CAS No. 61869-08-7 | Aladdin Scientific
Paroxetine – 97%, high purity , Serotonin transporter inhibitor, CAS No.61869-08-7, Serotonin transporter inhibitor
-
Paroxetine | CAS 61869-08-7 | AbMole BioScience
Paroxetine is a inhibitor of serotonin uptake. Buy 5-HT Receptor inhibitor Paroxetine




Reviews
There are no reviews yet.