Buy Quipazine (2-QP) Cas 4774-24-7
Buy Quipazine (2-QP) Cas 4774-24-7
Quipazine, also known as 1-(2-quinolinyl)piperazine (2-QP), is a serotonergic drug of the arylpiperazine family and an analogue of 1-(2-pyridinyl)piperazine which is used in scientific research.[2][3][4][5][6] It was first described in the 1960s and was originally intended as an antidepressant but was never developed or marketed for medical use.[2][7][5] The effects of quipazine in humans include nausea, vomiting, gastrointestinal disturbances, diarrhea, and, at higher doses, psychedelic effects.[2][1][4] Quipazine may represent the prototype of a novel structural class of psychedelic drugs.[2][8][9]
Use and effects
The effects and side effects of quipazine in humans have been described.[1][2] At a dose of 25 mg orally, they included nausea, flatulence, gastrointestinal discomfort, and diarrhea, with no LSD-like subjective effects.[1] Higher doses were not assessed due to serotonin 5-HT3 receptor-mediated side effects of nausea and gastrointestinal discomfort.[1][4] An anecdotal report in one or more subjects, in which the dose of quipazine was said to be 0.5 mg (sic), described quipazine as producing low-dose mescaline-like effects followed by onset of dysphoria and nausea.[1][2][10]
It was suggested by Jerrold C. Winter in 1994 that serotonin 5-HT3 receptor antagonists like ondansetron could allow for use of higher doses of quipazine and assessment of whether it produces clear psychedelic effects or not.[1] Alexander Shulgin subsequently reported in The Shulgin Index (2011), based on an anonymous report dated to 2007, that quipazine in combination with a serotonin 5-HT3 receptor antagonist, presumably ondansetron, produced a “full psychedelic response”.[4][11][2][12]
Interactions
Serotonin 5-HT3 receptor antagonists like ondansetron have been reported to block the nausea and vomiting induced by quipazine.[4][11][2][12] Serotonin 5-HT2A receptor antagonists like ketanserin have been reported to block the psychedelic-like effects of quipazine in animals.[2][4]
Pharmacology
Pharmacodynamics
| Target | Affinity (Ki, nM) |
|---|---|
| 5-HT1A | 230–>10,000 |
| 5-HT1B | 1,000 |
| 5-HT1D | 1,000–3,720 |
| 5-HT1E | ND |
| 5-HT1F | ND |
| 5-HT2A | 59–2,780 (Ki) 309 (EC50Tooltip half-maximal effective concentration) 62–71% (EmaxTooltip maximal efficacy) |
| 5-HT2B | 49–178 (Ki) 178 (EC50) 17% (Emax) |
| 5-HT2C | 54–1,344 (Ki) 339 (EC50) 57–69% (Emax) |
| 5-HT3 | 1.23–4.0 (Ki) 1.0 (EC50) ND (Emax) |
| 5-HT4 | >10,000 (guinea pig) |
| 5-HT5A | >10,000 (mouse) |
| 5-HT6 | 3,600 |
| 5-HT7 | 3,033 |
| α1 | >10,000 (rat) |
| α2 | 5,000 (rat) |
| β1 | 5,600 |
| β2 | 2,900 (rat) |
| D1 | >10,000 |
| D2 | >10,000 |
| D2-like | 3,920 (rat) |
| mAChTooltip Muscarinic acetylcholine receptor | >10,000 (rat) |
| TAAR1Tooltip Trace amine-associated receptor 1 | >10,000 (human) (EC50) |
| SERTTooltip Serotonin transporter | 30–143 |
| NETTooltip Norepinephrine transporter | ND |
| DATTooltip Dopamine transporter | ND |
| Notes: The smaller the value, the more avidly the drug binds to the site. All proteins are human unless otherwise specified. Refs: [14][15][13][16][17][18][19][20][21] | |
Quipazine is a serotonin 5-HT3 receptor agonist and to a lesser extent a serotonin 5-HT2A, 5-HT2B, and 5-HT2C receptor agonist as well as serotonin reuptake inhibitor.[2][3][17][14][15] It also shows affinity for serotonin 5-HT1 receptors, including the serotonin 5-HT1B receptor and to a lesser extent the serotonin 5-HT1A receptor.[22] Activation of the serotonin 5-HT3 is implicated in inducing nausea and vomiting as well as anxiety, which has limited the potential clinical usefulness of quipazine.[2][3][4]
Quipazine produces a head-twitch response and other psychedelic-consistent effects in animal studies including in mice, rats, and monkeys.[2][4][23][24][25] These effects appear to be mediated by activation of the serotonin 5-HT2A receptor, as they are blocked by serotonin 5-HT2A receptor antagonists like ketanserin.[2][4][25] The head twitches induced by quipazine are potentiated by the monoamine oxidase inhibitor (MAOI) pargyline.[25][26] Based on this, it has been suggested that quipazine may act as a serotonin releasing agent and that it may induce the head twitch response by a dual action of serotonin 5-HT2A receptor agonism and induction of serotonin release. Buy Quipazine (2-QP) Cas 4774-24-7
Besides the head-twitch response, quipazine fully substitutes for LSD and partially substitutes for mescaline in rodent drug discrimination tests.[1] In addition, quipazine substitutes for DOM in rodents and monkeys and this is blocked by serotonin 5-HT2A receptor antagonists like pizotyline and ketanserin.[2] When quipazine is used as the training drug, LSD, mescaline, and psilocybin all fully substitute for quipazine.[2] In monkeys, quipazine additionally produced LSD-like behavioral changes along with projectile vomiting.[1] In contrast to primates, rodents generally lack an emetic response, and hence the nausea and vomiting that quipazine can induce may not be a limiting factor in this order of animals.[2] Similarly to DOI, quipazine alters time perception in rodents.[27]
In addition to its psychedelic-like effects, quipazine can produce antiaggressive effects in rodents.[28] It can also produce tachycardia, including positive chronotropic and positive inotropic effects, through activation of the serotonin 5-HT3 receptor.[3]
Although quipazine does not generalize to dextroamphetamine in drug discrimination tests of dextroamphetamine-trained rodents, dextroamphetamine and cathinone have been found to partially generalize to quipazine in assays of quipazine-trained rodents.[29][30] In relation to this, it has been suggested that quipazine might possess some dopaminergic activity, as the discriminative stimulus properties of amphetamine appear to be mediated by dopamine signaling.[29][30] Relatedly, quipazine has been said to act as a dopamine receptor agonist in addition to serotonin receptor agonist.[25] Conversely however, the generalization may be due to serotonergic activities of amphetamine and cathinone.[31] Fenfluramine has been found to fully generalize to quipazine, but levofenfluramine, in contrast to quipazine, did not generalize to dextroamphetamine.[29][24]
Quipazine is said to differ in its pharmacology and effects from other serotonergic arylpiperazines like TFMPP and mCPP.[2][4] Relatedly, unlike quipazine, neither TFMPP nor mCPP substitute for DOM in drug discrimination tests.[2][4] In addition, DOM and TFMPP mutually antagonize each others’ stimulus effects.[2] In contrast to quipazine, TFMPP and mCPP show prominent bias or preference for the serotonin 5-HT2C receptor over the serotonin 5-HT2A receptor.[4]
Quipazine is a very weak agonist of the human trace amine-associated receptor 1 (TAAR1).[21]
Chemistry
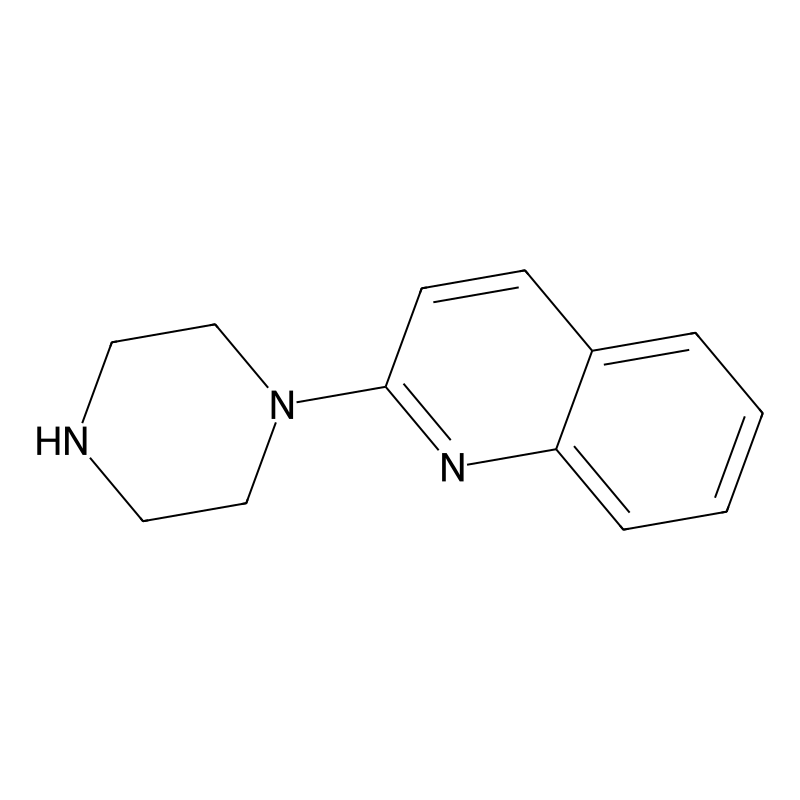
Quipazine
Content Navigation
CAS Number
Product Name
IUPAC Name
Molecular Formula
Molecular Weight
InChI
InChI Key
SMILES
Synonyms
Canonical SMILES
Investigating Serotonin Function:
- Serotonin Receptor Agonist: Quipazine acts as a non-selective serotonin receptor agonist, meaning it directly stimulates various types of serotonin receptors in the brain. This allows researchers to study the effects of serotonin activation on different neural processes .
- Modeling Serotonin-Related Disorders: By mimicking the effects of serotonin, quipazine can be used to model certain aspects of serotonin-related disorders like depression and anxiety in animal models .
Studying Neural Signaling:
- Head-Twitch Response: Quipazine induces a characteristic “head-twitch” response in rodents, which is a specific behavioral endpoint used to assess drug effects on serotonin signaling pathways .
- Investigating Gq/11 Signaling: Recent research suggests quipazine may influence a specific signaling pathway in the brain called the Gq/11 pathway, which is involved in various cellular functions .
Other Potential Applications:
- Antidepressant Potential: Quipazine was initially investigated as a potential antidepressant due to its serotonin-modulating effects; however, it was never developed for clinical use .
- Oxytocic Effects: Limited research suggests quipazine may also have oxytocic properties, meaning it could potentially stimulate uterine contractions, but further investigation is needed .
Quipazine is a compound belonging to the piperazine family, primarily recognized for its serotonergic activity. Initially developed as an antidepressant, it has garnered attention in scientific research due to its interaction with various serotonin receptors, particularly the 5-HT2A and 5-HT3 subtypes. Its chemical formula is C13H15N3, with a molar mass of approximately 213.284 g/mol . Despite its potential, quipazine was never fully developed for clinical use.
- Starting Materials: The primary reactants are 2-chloroquinoline and piperazine.
- Reaction Conditions: The reaction typically requires appropriate solvents and conditions that facilitate nucleophilic substitution.
- Purification: The product is purified through standard organic chemistry techniques such as recrystallization or chromatography .
Quipazine exhibits a range of biological activities, primarily as a serotonin reuptake inhibitor and a nonselective agonist at several serotonin receptors. It has been shown to produce significant effects in animal models, including:
- Psychedelic-like Effects: Quipazine induces head-twitch responses and other behaviors consistent with psychedelic activity in rodents and primates .
- Serotonin Receptor Binding: It binds effectively to 5-HT2A and 5-HT3 receptors, influencing various physiological responses such as smooth muscle contraction and neurotransmitter release .
While quipazine has demonstrated robust activity in preclinical studies, it failed to produce psychedelic effects in humans at doses up to 25 mg due to side effects associated with 5-HT3 receptor activation .
Quipazine’s applications are primarily confined to research settings:
Research has elucidated quipazine’s interactions with various serotonin receptors:
- 5-HT2A Receptor: Quipazine acts as an agonist, promoting signaling pathways associated with this receptor type. Studies indicate that it enhances inositol monophosphate accumulation, similar to classic psychedelics like 2,5-dimethoxy-4-iodoamphetamine (DOI) .
- Comparative Activity: Quipazine’s activity profile has been compared to other serotonergic compounds, revealing unique binding characteristics and effects on behavior in animal models .
Quipazine shares structural similarities with several other compounds within the piperazine class. Here are some notable comparisons:
Quipazine’s unique structure allows it to interact specifically with serotonin receptors while exhibiting a distinct pharmacological profile compared to these similar compounds. Its ability to act as both an agonist and a reuptake inhibitor sets it apart within the realm of serotonergic agents.
XLogP3
Hydrogen Bond Acceptor Count
Hydrogen Bond Donor Count
Exact Mass
Monoisotopic Mass
Heavy Atom Count
LogP
UNII
Related CAS
Other CAS
Wikipedia
Dates
Psychedelic-like Properties of Quipazine and Its Structural Analogues in Mice
Mario de la Fuente Revenga, Urjita H Shah, Nima Nassehi, Alaina M Jaster, Prithvi Hemanth, Salvador Sierra, Malgorzata Dukat, Javier González-Maeso
PMID: 33400504 DOI: 10.1021/acschemneuro.0c00291
Abstract
Known classic psychedelic serotonin 2A receptor (5-HT
R) agonists retain a tryptamine or phenethylamine at their structural core. However, activation of the 5-HT
R can be elicited by drugs lacking these fundamental scaffolds. Such is the case of the N-substituted piperazine quipazine. Here, we show that quipazine bound to and activated 5-HT
R as measured by [
H]ketanserin binding displacement, Ca
mobilization, and accumulation of the canonical G
signaling pathway mediator inositol monophosphate (IP
)
and
. Additionally, quipazine induced via 5-HT
R an expression pattern of immediate early genes (IEG) in the mouse somatosensory cortex consistent with that of classic psychedelics. In the mouse head-twitch response (HTR) model of psychedelic-like action, quipazine produced a lasting effect with high maximal responses during the peak effect that were successfully blocked by the 5-HT
R antagonist M100907 and absent in 5-HT
R knockout (KO) mice. The acute effect of quipazine on HTR appeared to be unaffected by serotonin depletion and was independent from 5-HT
R activation. Interestingly, some of these features were shared by its deaza bioisostere 2-NP, but not by other closely related piperazine congeners, suggesting that quipazine might represent a distinct cluster within the family of psychoactive piperazines. Together, our results add to the mounting evidence that quipazine’s profile matches that of classic psychedelic 5-HT
R agonists at cellular signaling and behavioral pharmacology levels.
Stimulation of 5-HT2A receptors recovers sensory responsiveness in acute spinal neonatal rats
Hillary E Swann, Sierra D Kauer, Jacob T Allmond, Michele R Brumley
PMID: 28004950 DOI: 10.1037/bne0000176
Abstract
Quipazine is a 5-HT
-receptor agonist that has been used to induce motor activity and promote recovery of function after spinal cord injury in neonatal and adult rodents. Sensory stimulation also activates sensory and motor circuits and promotes recovery after spinal cord injury. In rats, tail pinching is an effective and robust method of sacrocaudal sensory afferent stimulation that induces motor activity, including alternating stepping. In this study, responsiveness to a tail pinch following treatment with quipazine (or saline vehicle control) was examined in spinal cord transected (at midthoracic level) and intact neonatal rats. Rat pups were secured in the supine posture with limbs unrestricted. Quipazine or saline was administered intraperitoneally and after a 10-min period, a tail pinch was administered. A 1-min baseline period prior to tail-pinch administration and a 1-min response period postpinch was observed and hind-limb motor activity, including locomotor-like stepping behavior, was recorded and analyzed. Neonatal rats showed an immediate and robust response to sensory stimulation induced by the tail pinch. Quipazine recovered hind-limb movement and step frequency in spinal rats back to intact levels, suggesting a synergistic, additive effect of 5-HT-receptor and sensory stimulation in spinal rats. Although levels of activity in spinal rats were restored with quipazine, movement quality (high vs. low amplitude) was only partially restored. (PsycINFO Database Record
Cortico-reticulo-spinal circuit reorganization enables functional recovery after severe spinal cord contusion
Leonie Asboth, Lucia Friedli, Janine Beauparlant, Cristina Martinez-Gonzalez, Selin Anil, Elodie Rey, Laetitia Baud, Galyna Pidpruzhnykova, Mark A Anderson, Polina Shkorbatova, Laura Batti, Stephane Pagès, Julie Kreider, Bernard L Schneider, Quentin Barraud, Gregoire Courtine
PMID: 29556028 DOI: 10.1038/s41593-018-0093-5
Abstract
Severe spinal cord contusions interrupt nearly all brain projections to lumbar circuits producing leg movement. Failure of these projections to reorganize leads to permanent paralysis. Here we modeled these injuries in rodents. A severe contusion abolished all motor cortex projections below injury. However, the motor cortex immediately regained adaptive control over the paralyzed legs during electrochemical neuromodulation of lumbar circuits. Glutamatergic reticulospinal neurons with residual projections below the injury relayed the cortical command downstream. Gravity-assisted rehabilitation enabled by the neuromodulation therapy reinforced these reticulospinal projections, rerouting cortical information through this pathway. This circuit reorganization mediated a motor cortex-dependent recovery of natural walking and swimming without requiring neuromodulation. Cortico-reticulo-spinal circuit reorganization may also improve recovery in humans.
Quipazine Elicits Swallowing in the Arterially Perfused Rat Preparation: A Role for Medullary Raphe Nuclei?
Victor Bergé-Laval, Christian Gestreau
PMID: 32698469 DOI: 10.3390/ijms21145120
-
Buy Quipazine | 4774-24-7 | BenchChem
offers qualified products for CAS No. 4774-24-7 (Quipazine), please inquire us for more detail.
-
Quipazine | 4774-24-7 – MilliporeSigma
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
-
CAS 4774-24-7 Quipazine – Alfa Chemistry
Quipazine is a 5-HT agonist with a Ki value of 1.4 nM for displaces [3H]GR65630 from 5-HT3R in rat. Quipazine shows antiviral activity against SARS-CoV-2 with an EC50 of 31.64 μM. Quipazine …
quipazine – prefix CAS No. 4774-24-7 | Aladdin Scientific
quipazine , CAS No.4774-24-7, Agonist of 5-HT 2A receptor;Agonist of 5-HT 2B receptor;Agonist of 5-HT 2C receptor;Agonist of 5-HT 3A;Agonist of 5-HT 3AB
-
Quipazine | 4774-24-7 – ChemicalBook
Quipazine (4774-24-7) information like chemical properties,Structure,melting point,boiling point,density,molecular formula,molecular weight, physical …
-
4774-24-7 | MFCD00242605 | Quipazine
Quipazine. CAS Number: 4774-24-7. Catalog Number: AR00D8QN. MDL Number: MFCD00242605. Molecular Formula: C13H15N3. Molecular Weight: 213.2783.
-
4774-24-7 | Quipazine| 2-Piperazin-1-yl-quinoline;2- (piperazin-1-yl …
Quipazine is a piperazine-based nonselective serotonin (5-HT) receptor agonist with antidepressant and oxytocic activities.
-
CAS 4774-24-7 Quipazine – BOC Sciences
BOC Sciences is the world-leading provider of special chemicals. We offer qualified product Quipazine (4774-24-7), please inquire us for Quipazine (4774-24-7).
Buy Quipazine | 4774-24-7
Quipazine is a chemical compound used in scientific research.It possesses diverse applications, such as studying the effects of serotonin in the central nervous system.
-
4774-24-7 | Quipazine | ChemScene
ChemScene Provide CAS 4774-24-7, Quipazine, Formula:C13H15N3, MW:213.2800. We also provide services including custom synthesis, process optimization, commercial production, peptide custom …




Reviews
There are no reviews yet.