Buy Xorphanol mesylate (TR-5379) Cas 77287-89-9
Buy Xorphanol mesylate (TR-5379) Cas 77287-89-9
Xorphanol (INN; also known as xorphanol mesylate (USAN); developmental codes TR-5379 or TR-5379M) is an opioid analgesic of the morphinan family that was never marketed.[1][2][3]
Xorphanol is a mixed agonist–antagonist of opioid receptors,[4][5][6] acting preferentially as a high-efficacy partial agonist/near-full agonist of the κ-opioid receptor (Ki = 0.4 nM; EC50 = 3.3 nM; Imax = 49%; IA = 0.84)[7][8][9] and to a lesser extent as a partial agonist of the μ-opioid receptor (Ki = 0.25 nM; IC50 = 3.4 nM; Imax = 29%) with lower relative intrinsic activity and marked antagonistic potential (including the ability to antagonize morphine-induced effects and induce opioid withdrawal in opioid-dependent individuals).[3][10] The drug has also been found to act as an agonist of the δ-opioid receptor (Ki = 1.0 nM; IC50 = 8 nM; Imax = 76%). Buy Xorphanol mesylate (TR-5379) Cas 77287-89-9
Xorphanol produces potent analgesia, and was originally claimed to possess a minimal potential for dependence or abuse.[12][13][14] Moreover, side effects in animal studies were relatively mild, with only sedation and nausea being prominent, although it also produced convulsions at the highest dose tested.[15] However, human trials revealed additional side effects such as headaches and euphoria, and this was the subject of a lawsuit between the drug’s inventors and the company to which they had licensed the marketing rights, which claimed that these side effects had not been revealed to them during the license negotiations.[16] As a result of this dispute, the drug was never marketed commercially. Buy Xorphanol mesylate (TR-5379) Cas 77287-89-9
It has been encountered as a novel designer drug.[17]
See also
- 6-Methylenedihydrodesoxymorphine
- Butorphanol
- Cyclorphan
- Ketorfanol
- Levallorphan
- Levomethorphan
- Levorphanol
- Nalmefene
- Nalbuphine
- Nalorphine
- Oxilorphan
- Proxorphan
Xorphanol
Description
Structure
3D Structure
Properties
IUPAC Name |
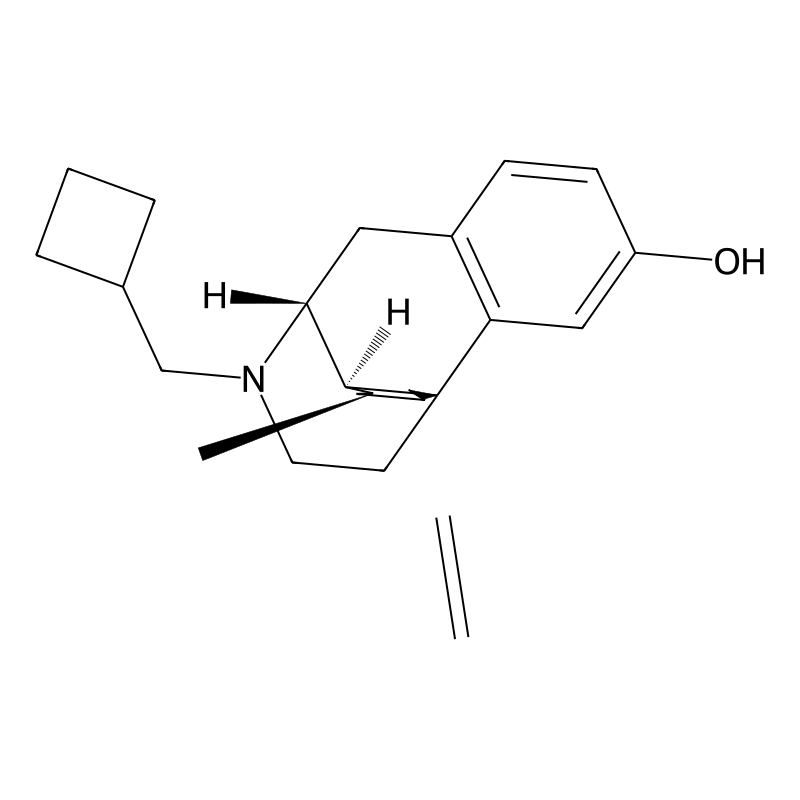
(1R,9R,10R,11S)-17-(cyclobutylmethyl)-11-methyl-13-methylidene-17-azatetracyclo[7.5.3.01,10.02,7]heptadeca-2(7),3,5-trien-4-ol | |
|---|---|---|
InChI |
InChI=1S/C23H31NO/c1-15-10-16(2)22-21-11-18-6-7-19(25)12-20(18)23(22,13-15)8-9-24(21)14-17-4-3-5-17/h6-7,12,16-17,21-22,25H,1,3-5,8-11,13-14H2,2H3/t16-,21+,22-,23+/m0/s1 | |
InChI Key |
AZJPPZHRNFQRRE-AZIXLERZSA-N | |
Canonical SMILES |
CC1CC(=C)CC23C1C(CC4=C2C=C(C=C4)O)N(CC3)CC5CCC5 | |
Isomeric SMILES |
C[C@H]1CC(=C)C[C@@]23[C@@H]1[C@@H](CC4=C2C=C(C=C4)O)N(CC3)CC5CCC5 | |
Molecular Formula |
C23H31NO | |
Molecular Weight |
337.5 g/mol | |
CAS No. |
77287-89-9 |
Foundational & Exploratory
Xorphanol’s Mechanism of Action: A Technical Guide
Xorphanol Receptor Binding Affinity: An In-depth Technical Guide
An In-Depth Technical Guide to the Chemical Structure and Pharmacology of Xorphanol
An In-depth Technical Guide to the Synthesis of Xorphanol Mesylate
An In-depth Technical Guide to the Solubility and Stability of Xorphanol
In Vitro Pharmacological Profile of Xorphanol: A Technical Guide
Neuropharmacological Profile of Xorphanol: An In-depth Technical Guide
Xorphanol’s Effect on the Central Nervous System: A Technical Guide
Molecular Targets of Xorphanol: An In-depth Technical Guide
The Genetic Basis of Xorphanol Response: A Technical Guide for Researchers and Drug Development Professionals
Methodological & Application
Application Notes and Protocols for the Use of Xorphanol in Cell Culture Assays
Application Notes and Protocols for the Dissolution of Xorphanol Mesylate for Injection
Xorphanol
Product Introduction
Xorphanol is classified as an opioid analgesic and is known for its activity at kappa opioid receptors. Its molecular formula is C23H31NO with a molecular weight of 337.5 g/mol. The compound is identified by the CAS number 77287-89-9 and has been documented in various chemical databases such as PubChem .
The synthesis of xorphanol involves multiple steps, beginning with the formation of the morphinan core structure. The general synthetic route includes:
- Formation of the Morphinan Core: This step involves cyclization reactions of suitable precursors to create the morphinan skeleton.
- Functionalization: Specific functional groups, including cyclobutylmethyl and methylene groups, are introduced at designated positions on the morphinan core.
- Purification: The final product undergoes purification processes such as recrystallization or chromatography to achieve high purity levels.
Although detailed industrial production methods are scarce due to xorphanol’s limited commercial use, standard principles of large-scale synthesis apply, focusing on optimizing reaction conditions and purification techniques.
Structural Data
- Molecular Formula: C23H31NO
- Molecular Weight: 337.5 g/mol
- CAS Number: 77287-89-9.
The compound features a complex arrangement typical of morphinan derivatives, contributing to its pharmacological activity.
Xorphanol participates in various chemical reactions, including:
- Oxidation: Xorphanol can be oxidized to yield ketones or carboxylic acids under appropriate conditions.
- Reduction: Reduction reactions can convert xorphanol into corresponding alcohols or amines.
- Substitution: Substitution reactions may occur where functional groups on the morphinan core are replaced with other substituents.
Common reagents used in these reactions include potassium permanganate for oxidation, lithium aluminum hydride for reduction, and alkyl halides for substitution.
Xorphanol primarily exerts its effects through interaction with kappa opioid receptors in the central nervous system. Upon binding to these receptors, xorphanol can modulate neurotransmitter release, leading to decreased pain perception and altered emotional responses to pain stimuli.
Data on Mechanism
Pharmacological studies indicate that xorphanol demonstrates significant binding affinity at kappa opioid receptors compared to other ligands, suggesting its potential efficacy as an analgesic agent .
Physical Properties
- Appearance: Typically appears as a white crystalline solid.
- Solubility: Soluble in organic solvents; specific solubility data may vary based on purity and formulation.
Chemical Properties
- Stability: Xorphanol is stable under standard storage conditions but may degrade under extreme pH or temperature conditions.
- Reactivity: Reacts with oxidizing agents and can undergo various transformations depending on the reaction environment.
Relevant analyses often include spectroscopic methods (e.g., NMR, IR) to confirm structural integrity and purity.
Xorphanol has been explored for various scientific applications, particularly in pharmacology as an analgesic agent. Its unique action at kappa opioid receptors makes it a candidate for further research in pain management therapies, especially in contexts where traditional opioids may pose risks of addiction or adverse effects.
Research continues into its pharmacological profiles and potential therapeutic uses, highlighting its significance within the field of medicinal chemistry .
Etymological Origins of the Nonproprietary Name “Xorphanol”
The nonproprietary name “Xorphanol” follows systematic conventions for opioid analgesics established by the World Health Organization (WHO) International Nonproprietary Name (INN) program. The prefix “Xor-” is linguistically unique and deliberately avoids similarities with existing opioid names (e.g., morphine, oxymorphone), serving both phonetic distinction and trademark purposes. The suffix “-phanol” is characteristic of synthetic morphinan derivatives, indicating its structural relationship to compounds like levorphanol and butorphanol [6] [8]. Chemically designated as N-(Cyclobutylmethyl)-8β-methyl-6-methylenemorphinan-3-ol, the name reflects:
- N-substitution: Cyclobutylmethyl group (contrasting with morphine’s methyl group)
- Skeletal modifications: 8β-methyl and 6-methylene groups on the morphinan core [6]
Table 1: Structural Elements of Xorphanol’s Nomenclature
| Name Component | Structural Feature | Pharmacological Significance |
|---|---|---|
| “Xor-“ | Unique identifier | Distinguishes from existing opioid classes |
| “-phanol” | Morphinan backbone | Indicates µ-opioid receptor affinity |
| Cyclobutylmethyl | N-substituent | Modifies receptor binding kinetics |
| 6-Methylene | C6 modification | Enhances κ-opioid receptor selectivity |
Developmental Trajectory in Opioid Analgesic Research (TR-5379/TR-5379M Codes)
Xorphanol emerged during the 1970–1980s push to develop analgesics with reduced abuse potential. Designated experimentally as TR-5379 (base compound) and TR-5379M (mesylate salt formulation USAN: xorphanol mesylate), it represented a strategic shift toward mixed agonist-antagonist opioids [1] [6]. Key developmental milestones:
- Molecular Design: Engineered as a morphinan derivative with cyclobutylmethyl substitution and methyl/methylene modifications to alter receptor interaction dynamics versus classical opioids like morphine [6] [7]
- Mechanistic Differentiation: Early studies identified its κ-opioid receptor (KOR) partial agonism (Ki = 0.4 nM) combined with µ-opioid receptor (MOR) partial agonism/antagonism (Ki = 0.25 nM), a profile hypothesized to limit respiratory depression and dependence [6] [7]
- Analgesic Validation: Preclinical studies demonstrated potent analgesia in animal models, with oral bioavailability and duration supporting clinical potential [1]
Table 2: Xorphanol’s Receptor Binding Profile
| Receptor Type | Binding Affinity (Ki) | Intrinsic Activity | Functional Role |
|---|---|---|---|
| κ-opioid (KOR) | 0.4 nM | Partial agonist (49% Imax) | Primary analgesic pathway |
| µ-opioid (MOR) | 0.25 nM | Partial agonist (29% Imax) | Analgesia with antagonistic potential |
| δ-opioid (DOR) | 1.0 nM | Agonist (76% Imax) | Modulatory effects |
Properties
CAS Number
Product Name
IUPAC Name
Molecular Formula
Molecular Weight
InChI
InChI Key
SMILES
Solubility
Synonyms
N-CBM-MMM
TR 5379
TR-5379M
xorphanol mesylate
Canonical SMILES
Isomeric SMILES
Xorphanol
Content Navigation
CAS Number
Product Name
IUPAC Name
Molecular Formula
Molecular Weight
InChI
InChI Key
SMILES
solubility
Synonyms
Canonical SMILES
Isomeric SMILES
Xorphanol is a synthetic compound classified as a mixed agonist-antagonist of opioid receptors, primarily exhibiting high-efficacy partial agonist activity at the kappa-opioid receptor. It is structurally related to morphinan derivatives and is known for its analgesic properties. Xorphanol has gained attention for its unique pharmacological profile, which allows it to provide effective pain relief while minimizing some of the adverse effects commonly associated with traditional opioid medications, such as respiratory depression and dependence .
- Reduction Reactions: Xorphanol can be reduced to form corresponding alcohols or amines, which may alter its pharmacological properties .
- Substitution Reactions: The compound can undergo nucleophilic substitution, allowing for modifications that can enhance its therapeutic efficacy or alter its receptor binding characteristics .
Xorphanol exhibits notable biological activity as an analgesic agent. Its mechanism of action involves:
- Opioid Receptor Interaction: Xorphanol preferentially activates kappa-opioid receptors while displaying antagonistic properties at mu-opioid receptors. This selective activity contributes to its analgesic effects without the full spectrum of side effects seen with mu-opioid agonists .
- Resistance to Antagonism: In vitro studies have indicated that xorphanol possesses anti-naloxone properties, meaning it can resist antagonism by traditional opioid antagonists, which may enhance its therapeutic potential in certain clinical contexts .
The synthesis of xorphanol typically involves multi-step organic reactions starting from readily available precursors. Common methods include:
- Starting Materials: Utilizing morphinan derivatives as the starting point.
- Functional Group Modifications: Employing techniques such as alkylation and reduction to introduce necessary functional groups.
- Purification: The final product is purified through crystallization or chromatography techniques to ensure high purity suitable for pharmaceutical applications.
These methods allow for the production of xorphanol in a laboratory setting, making it accessible for research and clinical use .
Xorphanol is primarily utilized in the medical field for:
Studies investigating xorphanol’s interactions with other substances have revealed significant insights:
- Opioid Receptor Dynamics: Research has shown that xorphanol’s interaction with kappa-opioid receptors can modulate pain pathways differently than traditional opioids, potentially leading to fewer side effects .
- Drug Interactions: Clinical evaluations have indicated that xorphanol may interact with other central nervous system depressants, necessitating careful monitoring when co-administered with such drugs.
These studies highlight xorphanol’s unique position within the opioid pharmacology landscape.
Xorphanol shares structural and functional similarities with several other compounds in the opioid category. Here are some notable comparisons:
| Compound Name | Type | Primary Action | Unique Feature |
|---|---|---|---|
| Morphine | Full Agonist | Strong mu-opioid receptor agonist | High potential for dependence and respiratory depression |
| Buprenorphine | Partial Agonist | Mu-opioid receptor partial agonist | Ceiling effect on respiratory depression |
| Nalbuphine | Mixed Agonist/Antagonist | Kappa agonist, mu antagonist | Less addictive potential compared to full agonists |
| Pentazocine | Mixed Agonist | Kappa agonist | Can cause dysphoria in some patients |
Xorphanol’s unique efficacy at the kappa-opioid receptor combined with its resistance to naloxone antagonism distinguishes it from these compounds, making it a promising candidate for further research and clinical application .
Synthetic Pathways and Methodologies
The synthesis of Xorphanol, a morphinan alkaloid derivative with molecular formula C₂₃H₃₁NO, follows established synthetic methodologies for morphinan alkaloids while incorporating specific structural modifications unique to this compound [1] [2]. The synthetic approach to Xorphanol utilizes several key methodologies that have been developed for morphinan alkaloid synthesis over the past decades.
The primary synthetic pathway begins with the construction of the phenanthrene-based core structure, which can be achieved through multiple routes. The most efficient approaches include the use of intramolecular Diels-Alder cycloaddition reactions, which have proven highly effective for constructing the complex polycyclic framework of morphinan alkaloids [3] [4]. This methodology allows for the stereoselective formation of the required ring system with appropriate substitution patterns.
Alternative synthetic methodologies include the biomimetic oxidative phenol coupling approach, which mimics the natural biosynthetic pathway observed in opium poppy [5] [6]. This approach involves the oxidative coupling of phenolic precursors to form the characteristic morphinan core structure. The coupling reaction typically employs oxidizing agents such as ferric chloride or electrochemical oxidation conditions to achieve the desired phenol-phenol coupling [7].
Recent advances in synthetic methodology have demonstrated the utility of anodic aryl-aryl coupling reactions for morphinan synthesis [7]. These electrochemical approaches offer advantages in terms of selectivity and environmental considerations, as they avoid the use of stoichiometric oxidizing agents. The electrochemical coupling reactions can be performed under mild conditions while maintaining high stereoselectivity.
Formation of the Morphinan Core Structure
The morphinan core structure of Xorphanol is characterized by its tetracyclic framework consisting of a phenanthrene moiety fused to a piperidine ring [8]. The formation of this core structure represents one of the most challenging aspects of Xorphanol synthesis due to the complex stereochemistry and the need for precise regioselectivity.
The construction of the morphinan core typically proceeds through a series of cyclization reactions. The initial step involves the formation of the phenanthrene system, which can be achieved through various approaches including the Diels-Alder reaction of appropriately substituted benzofuran derivatives with diene partners [9]. This reaction provides the basic aromatic framework with the correct regiochemistry for subsequent transformations.
The key cyclization step involves the formation of the nitrogen-containing ring system. This transformation typically employs intramolecular cyclization reactions, such as the Pictet-Spengler reaction or related acid-catalyzed cyclizations [8]. The cyclization must be carefully controlled to ensure the correct stereochemistry at the newly formed chiral centers.
The formation of the morphinan core also requires careful consideration of the substitution pattern. The specific positioning of functional groups must be established early in the synthesis to ensure proper reactivity for subsequent transformations. This includes the placement of hydroxyl groups, methoxy groups, and other substituents that define the final structure of Xorphanol.
Stereochemical Considerations in Synthesis
Xorphanol contains multiple chiral centers within its morphinan framework, requiring careful stereochemical control throughout the synthetic sequence [10]. The compound exhibits specific stereochemistry that must be precisely controlled to ensure the desired pharmacological properties.
The stereoc
-
Buy Xorphanol | 77287-89-9 | >98% – Smolecule
Xorphanol is a synthetic compound classified as a mixed agonist-antagonist of opioid receptors, primarily exhibiting high-efficacy partial agonist activity at the kappa-opioid receptor. It is structurally …
-
Buy Xorphanol (EVT-286151) | 77287-89-9 – evitachem.com
EvitaChem offers qualified products for Xorphanol (evt-286151), please inquire us for more detail.
-
Buy Xorphanol | 77287-89-9 | BenchChem
Benchchem offers qualified products for CAS No. 77287-89-9 (Xorphanol), please inquire us for more detail.
-
Xorphanol | bioactive compound | CAS# 77287-89-9 | InvivoChem
Xorphanol is a novel and potent bioactive compound Physicochemical Properties Molecular Formula C23H31NO Molecular Weight 337.507 Exact Mass 337.241 CAS # 77287-89-9 PubChem CID …
-
CAS 77287-89-9 Xorphanol – BOC Sciences
BOC Sciences is the world-leading provider of special chemicals. We offer qualified product Xorphanol (77287-89-9), please inquire us for Xorphanol (77287-89-9).
-
Reviews: 1
-
-
77287-89-9 | Xorphanol | MOLCORE
MolCore offers CAS No.77287-89-9, Xorphanol for your process needs.Find product specific information including MFCD00864208,77287-89-9 MSDS,Price,Custom Synthesis.
-
Reviews: 33
-
-
Xorphanol – TargetMol
All types of co-solvents required for the protocol, such as DMSO, PEG300 / PEG400, Tween 80, SBE-β-CD, corn oil are available for purchase on the TargetMol website with a simple click.
-
Reviews: 1
-
-
Xorphanol mesylate;TR 5379M;TR-5379M;TR5379M 77287-90-2 …
Jan 1, 2022 · Xorphanol mesylate;TR 5379M;TR-5379M;TR5379M,,/,cas No:77287-90-2,Formula:C24H35NO4S,purity:- TargetMol Chemicals Inc.
-
Welcome to Hodoodo Chemicals
WARNING: This product is for research use only, not for human or veterinary use. Hodoodo CAT#: H341453 CAS#: 77287-89-9
-
Xorphanol | 77287-89-9
Xorphanol (77287-89-9) information like chemical properties,Structure,melting point,boiling point,density,molecular formula,molecular weight, physical …





Reviews
There are no reviews yet.