Buy Zenazocine (WIN-42964) Cas 74559-85-6
Buy Zenazocine (WIN-42964) Cas 74559-85-6
Zenazocine (INN; WIN-42,964) is an opioid analgesic of the benzomorphan family which made it to phase II clinical trials before development was ultimately halted and it was never marketed.[1][2] It acts as a partial agonist of the μ- and δ-opioid receptors, with less intrinsic activity at the former receptor and more at the latter receptor (hence, it behaves more antagonistically at the former and more agonistically at the latter), and produces antinociceptive effects in animal studies. Buy Zenazocine (WIN-42964) Cas 74559-85-6
See also
Zenazocine mesylate is a synthetic compound primarily classified as a non-selective opioid receptor agonist. It has garnered attention in pharmacological research due to its unique properties and potential applications in pain management and other therapeutic areas. The compound is derived from the modification of existing opioid structures, aiming to enhance efficacy while minimizing adverse effects associated with traditional opioids. Buy Zenazocine (WIN-42964) Cas 74559-85-6
Zenazocine mesylate is synthesized in laboratory settings, utilizing various chemical precursors and reagents. The compound’s development is rooted in the need for effective analgesics that can overcome the limitations of conventional opioids, such as tolerance and addiction. Buy Zenazocine (WIN-42964) Cas 74559-85-6, Buy Zenazocine (WIN-42964) Cas 74559-85-6, Buy Zenazocine (WIN-42964) Cas 74559-85-6, Buy Zenazocine (WIN-42964) Cas 74559-85-6, Buy Zenazocine (WIN-42964) Cas 74559-85-6, Buy Zenazocine (WIN-42964) Cas 74559-85-6, Buy Zenazocine (WIN-42964) Cas 74559-85-6, Buy Zenazocine (WIN-42964) Cas 74559-85-6, Buy Zenazocine (WIN-42964) Cas 74559-85-6, Buy Zenazocine (WIN-42964) Cas 74559-85-6, Buy Zenazocine (WIN-42964) Cas 74559-85-6
Zenazocine mesylate is classified under the category of opioid analgesics. It interacts with multiple opioid receptors, including the mu, delta, and kappa receptors, which are known to mediate pain relief and other physiological responses.
Methods
The synthesis of Zenazocine mesylate involves several key steps, typically starting from a precursor compound that undergoes various chemical transformations. The general synthetic route includes:
- Formation of the Core Structure: The initial step involves the construction of the central phenanthrene or morphinan skeleton, which serves as the backbone for the compound.
- Modification with Functional Groups: Subsequent reactions introduce specific functional groups that enhance receptor binding affinity and selectivity.
- Mesylation: The final step involves the introduction of the mesylate group, which is crucial for improving solubility and pharmacokinetic properties.
Technical Details
The synthesis may utilize techniques such as:
- Refluxing: To facilitate reactions at elevated temperatures.
- Chromatography: For purification of intermediates and final products.
- Spectroscopic Methods: Including Nuclear Magnetic Resonance (NMR) and Mass Spectrometry (MS) for structural confirmation.
Structure
The molecular structure of Zenazocine mesylate can be represented by its chemical formula, which includes carbon, hydrogen, nitrogen, oxygen, and sulfur atoms. The specific arrangement of these atoms defines its pharmacological properties.
Data
- Molecular Formula: CxHyNzOwSz (exact values depend on specific substitutions).
- Molecular Weight: Approximately 400-500 g/mol (varies based on structure).
- 3D Structure: Can be visualized using molecular modeling software to understand its interaction with opioid receptors.
Reactions
Zenazocine mesylate can participate in various chemical reactions typical of opioid compounds:
- Hydrolysis: In aqueous environments, leading to the breakdown of the mesylate group.
- Oxidation/Reduction: Potential modifications that alter its activity profile.
- Substitution Reactions: Where functional groups can be exchanged or modified to enhance efficacy.
Technical Details
The reactivity of Zenazocine mesylate is influenced by its functional groups and steric hindrance within its structure. These factors play a significant role in determining its stability and interaction with biological systems.
Process
Zenazocine mesylate exerts its effects primarily through agonistic activity at opioid receptors. The mechanism involves:
- Receptor Binding: Zenazocine mesylate binds to mu-opioid receptors in the central nervous system.
- Signal Transduction: This binding activates intracellular signaling pathways, primarily involving G-proteins.
- Physiological Response: The activation leads to decreased neurotransmitter release (e.g., substance P), resulting in analgesia.
Data
Studies have shown that Zenazocine mesylate has a binding affinity comparable to other potent opioids but with a distinct profile that may reduce side effects such as respiratory depression.
Physical Properties
- Appearance: Typically exists as a white crystalline powder.
- Solubility: Soluble in organic solvents like ethanol; limited solubility in water due to hydrophobic characteristics.
- Melting Point: Specific melting point data may vary but often falls within a range typical for similar compounds.
Chemical Properties
- Stability: Generally stable under standard laboratory conditions but sensitive to light and moisture.
- pH Sensitivity: May exhibit changes in solubility or stability based on pH levels.
Scientific Uses
Zenazocine mesylate is primarily studied for its potential applications in:
- Pain Management: As an analgesic agent for acute and chronic pain conditions.
- Research Models: Utilized in pharmacological studies to better understand opioid receptor interactions and develop safer analgesics.
- Therapeutic Development: Investigated as a candidate for formulations aimed at reducing opioid dependence while maintaining effective pain relief.
Discovery and Development Within the Benzomorphan Analogue Series
Zenazocine Mesylate originated from systematic structural modifications of the benzomorphan scaffold, a class of compounds first investigated in the 1960s for their potent analgesic properties with reduced addiction potential compared to traditional morphinan opioids. The benzomorphan nucleus is characterized by a partially reduced phenanthrene structure with a dimethylaminoethyl side chain, creating a distinctive tricyclic framework that enables diverse pharmacological activities depending on specific substituents. Zenazocine was synthesized as part of a molecular optimization program targeting compounds with mixed agonist-antagonist activity at opioid receptors [1] [8].
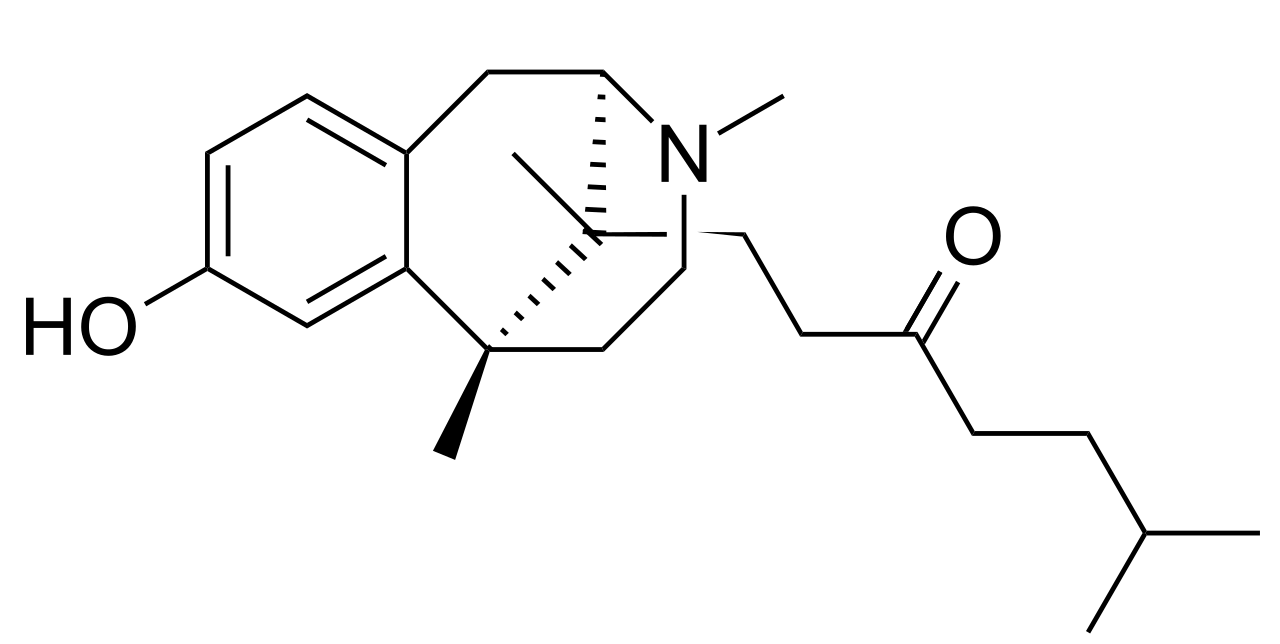
The molecular structure of Zenazocine (chemical name: 1-[(1S,9R)-4-hydroxy-1,10,13-trimethyl-10-azatricyclo[7.3.1.02,7]trideca-2,4,6-trien-13-yl]-6-methyl-3-heptanone) features critical benzomorphan elements including the characteristic aromatic ring and piperidine nitrogen, while incorporating a unique 6-methyl-3-heptanone side chain that distinguished it from earlier analogues like pentazocine and cyclazocine. The mesylate salt (methanesulfonate salt) was developed to improve solubility and crystallinity for pharmaceutical processing, with the salt form having the chemical formula C₂₃H₃₅NO₂·CH₄O₃S and molecular weight of 453.635 g/mol [3] [8].
Table 1: Chemical Profile of Zenazocine Mesylate
| Property | Specification |
|---|---|
| Chemical Formula | C₂₃H₃₅NO₂·CH₄O₃S |
| Molecular Weight | 453.635 g/mol |
| Parent Compound CAS | 68681-43-6 |
| Mesylate Salt CAS | 74559-85-6 |
| Stereochemistry | Racemic mixture |
| Defined Stereocenters | 3 chiral centers |
| Lipophilicity (LogP) | 4.71 (predictive) |
| Development Codes | WIN-42,964; WIN-42964-4; NIH-9625; MCV-4176 |
The development pathway reflected a deliberate shift from pure μ-opioid receptor agonists toward compounds with mixed receptor activities. Early pharmacological screening demonstrated that Zenazocine Mesylate exhibited the targeted mixed agonist-antagonist profile, producing antinociceptive effects in animal models while potentially reducing abuse liability compared to full μ-agonists. This promising preclinical profile supported advancement to human clinical trials during the late 1970s and early 1980s, where it underwent Phase I safety evaluation and Phase II efficacy assessment before development termination [1] [3].
Structural Classification as a Mixed Opioid Agonist-Antagonist
Zenazocine Mesylate belongs to the benzomorphan subclass of opioids characterized by a partially reduced phenanthrene core structure with a dimethylaminoethyl side chain. This specific structural framework enables distinctive interactions with opioid receptor subtypes that differ from those of morphinan-derived opioids or fully synthetic piperidine derivatives. The compound exists as a racemic mixture containing both enantiomers, with the (2R,6S,11S) configuration identified as the biologically active form responsible for opioid receptor interactions [3] [8].
Pharmacological characterization revealed that Zenazocine functions as a partial agonist at the μ-opioid receptor (MOR) with low intrinsic activity, producing limited analgesic efficacy through this pathway while simultaneously acting as a partial agonist at the δ-opioid receptor (DOR) with comparatively higher intrinsic activity. This dual activity profile positioned Zenazocine Mesylate pharmacologically as a mixed agonist-antagonist, with predominant antagonistic behavior at the MOR and agonistic activity at the DOR. The differential intrinsic activity at these receptors distinguished it from earlier benzomorphans and contributed to its unique pharmacological effects [1].
Table 2: Comparative Receptor Binding Profile of Benzomorphan Opioids
| Compound | μ-Opioid Receptor Activity | δ-Opioid Receptor Activity | κ-Opioid Receptor Activity |
|---|---|---|---|
| Zenazocine | Partial agonist (low IA) | Partial agonist (moderate IA) | Weak/negligible affinity |
| Pentazocine | Partial agonist/antagonist | Weak antagonist | Agonist |
| Cyclazocine | Antagonist | Weak antagonist | Agonist |
| Phenazocine | Full agonist | Weak agonist | Weak agonist |
The structural basis for this mixed activity profile resides in specific molecular features: the 4-hydroxy aromatic ring provides essential hydrogen bonding capacity for receptor interaction, while the 6-methyl-3-heptanone side chain creates steric and electronic properties that modulate receptor activation efficacy. Molecular modeling suggests that the extended hydrophobic side chain impedes optimal positioning within the MOR activation pocket while allowing productive engagement with DOR conformations associated with partial agonism. This differential engagement underlies the compound’s ability to produce antinociception primarily through DOR-mediated pathways while antagonizing full MOR agonists [1] [8].
Termination of Clinical Development: Scientific and Methodological Factors
Zenazocine Mesylate reached Phase II clinical trials for pain management indications before development was permanently halted. Analysis of available evidence indicates termination resulted from multifactorial challenges spanning pharmacological limitations, clinical trial methodology constraints, and evolving developmental priorities rather than isolated safety concerns [1] [3].
The primary scientific factor contributing to discontinuation involved inadequate therapeutic efficacy relative to established analgesics. Despite promising antinociceptive effects in animal models, Zenazocine Mesylate demonstrated insufficient analgesic efficacy in human trials across multiple pain models. This efficacy deficit likely stemmed from its suboptimal intrinsic activity profile at opioid receptors—specifically, the low intrinsic activity at the μ-opioid receptor combined with partial δ-agonism failed to produce consistently robust analgesia comparable to full MOR agonists. Additionally, its physicochemical properties, particularly the molecular weight (453.635 g/mol) and lipophilicity (LogP ~4.71), potentially limited central nervous system penetration across the blood-brain barrier, restricting access to central opioid receptors critical for analgesic efficacy [2] [9].
Methodological factors in clinical development further contributed to discontinuation. Phase II trial designs contemporary to Zenazocine’s development often employed conventional analgesic assessments insufficiently sensitive to detect partial agonist efficacy. Additionally, the compound’s development coincided with increasing recognition that drug tissue exposure and selectivity critically influence clinical outcomes—factors not adequately addressed in early-phase trials. The emerging Structure-Tissue Exposure/Selectivity-Activity Relationship (STAR) framework suggests Zenazocine may have been classified as a Class II agent (high specificity but low tissue exposure/selectivity) or Class IV agent (low specificity and low tissue exposure), both associated with high failure risk due to either insufficient efficacy or unacceptable toxicity profiles at therapeutic doses [2] [7].
The broader context of developmental attrition further contextualizes Zenazocine’s discontinuation. Contemporary analyses indicate approximately 90% of drug candidates entering clinical development ultimately fail, with 40-50% failing due to inadequate efficacy and 30% due to safety concerns. Opioid development faces particular challenges, with Phase II failure rates exceeding 70% and Phase III failure approaching 50%, significantly higher than non-CNS therapeutic areas. Zenazocine Mesylate’s termination exemplifies these industry-wide challenges in developing centrally-acting agents with complex pharmacological profiles [4] [7].
Zenazocine
- Chemical Name:Zenazocine
- CAS No.:68681-43-6
- Molecular Formula:C23H35NO2
- Molecular Weight:357.534
- Hs Code.:
- Mol file:68681-43-6.mol
Synonyms:
- Business phase:
- The product has achieved commercial mass production*data from LookChem market partment
- Manufacturers and distributors:
-
- Manufacture/Brand
- Chemicals and raw materials
- Packaging
- price
- American Custom Chemicals Corporation
- ZENAZOCINE 95.00%
- 5MG
- $ 501.63
- PSA:40.54000
- LogP:4.63980
- Purity/Quality:
- 99% *data from raw suppliers
ZENAZOCINE 95.00% *data from reagent suppliers
- Pictogram(s):
- Hazard Codes:
- MSDS Files:
There total 2 articles about Zenazocine which guide to synthetic route it. The literature collected by LookChem mainly comes from the sharing of users and the free literature resources found by Internet computing technology. We keep the original model of the professional version of literature to make it easier and faster for users to retrieve and use. At the same time, we analyze and calculate the most feasible synthesis route with the highest yield for your reference as below:
Zenazocine mesylate (USAN) (CAS No. 74559-85-6) SDS
- CAS No: 74559-85-6
- Molecular Weight: 453.63500
- Molecular Formula: C24H39NO5S
Names and IdentifiersPropertiesSafety and HandlingComputational chemical data
SAFETY DATA SHEETS
According to Globally Harmonized System of Classification and Labelling of Chemicals (GHS) – Sixth revised edition
Version: 1.0
Creation Date: Aug 15, 2017
Revision Date: Aug 15, 2017
1.Identification
1.1GHS Product identifier
| Product name | Zenazocine mesylate (USAN) |
|---|
1.2Other means of identification
| Product number | – |
|---|---|
| Other names | UNII-8WIL6L9U01 |
1.3Recommended use of the chemical and restrictions on use
| Identified uses | For industry use only. |
|---|---|
| Uses advised against | no data available |
1.4Supplier’s details
| Company | XiXisys.com |
|---|---|
| Address | XiXisys.com |
| Telephone | XiXisys.com |
| Fax | XiXisys.com |
1.5Emergency phone number
| Emergency phone number | – |
|---|---|
| Service hours | Monday to Friday, 9am-5pm (Standard time zone: UTC/GMT +8 hours). |
2.Hazard identification
2.1Classification of the substance or mixture
no data available
2.2GHS label elements, including precautionary statements
| Pictogram(s) | no data available |
|---|---|
| Signal word | no data available |
| Hazard statement(s) | no data available |
| Precautionary statement(s) | |
| Prevention | no data available |
| Response | no data available |
| Storage | no data available |
| Disposal | no data available |
2.3Other hazards which do not result in classification
no data available
3.Composition/information on ingredients
3.1Substances
| Chemical name | Common names and synonyms | CAS number | EC number | Concentration |
|---|---|---|---|---|
| Zenazocine mesylate (USAN) | Zenazocine mesylate (USAN) | 74559-85-6 | none | 100% |
4.First-aid measures
4.1Description of necessary first-aid measures
General advice
Consult a physician. Show this safety data sheet to the doctor in attendance.
If inhaled
If breathed in, move person into fresh air. If not breathing, give artificial respiration. Consult a physician.
In case of skin contact
Wash off with soap and plenty of water. Consult a physician.
In case of eye contact
Rinse thoroughly with plenty of water for at least 15 minutes and consult a physician.
If swallowed
Never give anything by mouth to an unconscious person. Rinse mouth with water. Consult a physician.
4.2Most important symptoms/effects, acute and delayed
no data available
4.3Indication of immediate medical attention and special treatment needed, if necessary
no data available
5.Fire-fighting measures
5.1Extinguishing media
Suitable extinguishing media
Use water spray, alcohol-resistant foam, dry chemical or carbon dioxide.
5.2Specific hazards arising from the chemical
no data available
5.3Special protective actions for fire-fighters
Wear self-contained breathing apparatus for firefighting if necessary.
6.Accidental release measures
6.1Personal precautions, protective equipment and emergency procedures
Use personal protective equipment. Avoid dust formation. Avoid breathing vapours, mist or gas. Ensure adequate ventilation. Evacuate personnel to safe areas. Avoid breathing dust. For personal protection see section 8.
6.2Environmental precautions
Prevent further leakage or spillage if safe to do so. Do not let product enter drains. Discharge into the environment must be avoided.
6.3Methods and materials for containment and cleaning up
Pick up and arrange disposal. Sweep up and shovel. Keep in suitable, closed containers for disposal.
7.Handling and storage
7.1Precautions for safe handling
Avoid contact with skin and eyes. Avoid formation of dust and aerosols. Avoid exposure – obtain special instructions before use.Provide appropriate exhaust ventilation at places where dust is formed. For precautions see section 2.2.
7.2Conditions for safe storage, including any incompatibilities
Store in cool place. Keep container tightly closed in a dry and well-ventilated place.
8.Exposure controls/personal protection
8.1Control parameters
Occupational Exposure limit values
no data available
Biological limit values
no data available
8.2Appropriate engineering controls
Handle in accordance with good industrial hygiene and safety practice. Wash hands before breaks and at the end of workday.
8.3Individual protection measures, such as personal protective equipment (PPE)
Eye/face protection
Safety glasses with side-shields conforming to EN166. Use equipment for eye protection tested and approved under appropriate government standards such as NIOSH (US) or EN 166(EU).
Skin protection
Wear impervious clothing. The type of protective equipment must be selected according to the concentration and amount of the dangerous substance at the specific workplace. Handle with gloves. Gloves must be inspected prior to use. Use proper glove removal technique(without touching glove’s outer surface) to avoid skin contact with this product. Dispose of contaminated gloves after use in accordance with applicable laws and good laboratory practices. Wash and dry hands. The selected protective gloves have to satisfy the specifications of EU Directive 89/686/EEC and the standard EN 374 derived from it.
Respiratory protection
Wear dust mask when handling large quantities.
Thermal hazards
no data available
9.Physical and chemical properties
| Physical state | no data available |
|---|---|
| Colour | no data available |
| Odour | no data available |
| Melting point/ freezing point | no data available |
| Boiling point or initial boiling point and boiling range | no data available |
| Flammability | no data available |
| Lower and upper explosion limit / flammability limit | no data available |
| Flash point | no data available |
| Auto-ignition temperature | no data available |
| Decomposition temperature | no data available |
| pH | no data available |
| Kinematic viscosity | no data available |
| Solubility | no data available |
| Partition coefficient n-octanol/water (log value) | no data available |
| Vapour pressure | no data available |
| Density and/or relative density | no data available |
| Relative vapour density | no data available |
| Particle characteristics | no data available |
10.Stability and reactivity
10.1Reactivity
no data available
10.2Chemical stability
Stable under recommended storage conditions.
10.3Possibility of hazardous reactions
no data available
10.4Conditions to avoid
no data available
10.5Incompatible materials
no data available
10.6Hazardous decomposition products
no data available
11.Toxicological information
Acute toxicity
- Oral: no data available
- Inhalation: no data available
- Dermal: no data available
Skin corrosion/irritation
no data available
Serious eye damage/irritation
no data available
Respiratory or skin sensitization
no data available
Germ cell mutagenicity
no data available
Carcinogenicity
no data available
Reproductive toxicity
no data available
STOT-single exposure
no data available
STOT-repeated exposure
no data available
Aspiration hazard
no data available
12.Ecological information
12.1Toxicity
- Toxicity to fish: no data available
- Toxicity to daphnia and other aquatic invertebrates: no data available
- Toxicity to algae: no data available
- Toxicity to microorganisms: no data available
12.2Persistence and degradability
no data available
12.3Bioaccumulative potential
no data available
12.4Mobility in soil
no data available
12.5Other adverse effects
no data available
13.Disposal considerations
13.1Disposal methods
Product
The material can be disposed of by removal to a licensed chemical destruction plant or by controlled incineration with flue gas scrubbing. Do not contaminate water, foodstuffs, feed or seed by storage or disposal. Do not discharge to sewer systems.
Contaminated packaging
Containers can be triply rinsed (or equivalent) and offered for recycling or reconditioning. Alternatively, the packaging can be punctured to make it unusable for other purposes and then be disposed of in a sanitary landfill. Controlled incineration with flue gas scrubbing is possible for combustible packaging materials.
Table 3: Clinical Development Failure Factors Relevant to Zenazocine Mesylate
| Failure Factor Category | Specific Challenges | Applicability to Zenazocine |
|---|---|---|
| Efficacy Limitations | Suboptimal intrinsic activity at target receptors | Low intrinsic activity at μ-opioid receptor |
| Inadequate tissue exposure/selectivity | Limited CNS penetration predicted | |
| Trial Methodology | Insensitive efficacy endpoints | Conventional pain assessments in Phase II |
| Inadequate pharmacodynamic biomarkers | Limited receptor occupancy data |
-
Buy Zenazocine mesylate (EVT-10914219) | 74559-85-6
Zenazocine mesylate is a synthetic compound primarily classified as a non-selective opioid receptor agonist. It has garnered attention in pharmacological research due to its unique properties and …
-
China Low Price Zenazocine Mesylate (USAN) Manufacturers, …
As one of the leading zenazocine mesylate (usan) manufacturers and suppliers in China, we warmly welcome you to wholesale brain and nervous system at low price from our online mall!
-
Zenazocine mesylate (USAN) | CAS#:74559-85-6 | Chemsrc
Chemsrc provides Zenazocine mesylate (USAN) (CAS#:74559-85-6) MSDS, density, melting point, boiling point, structure, formula, molecular weight etc. Articles of Zenazocine mesylate (USAN) are …
-
Zenazocine mesylate (USAN), CasNo.74559-85-6 Zibo Hangyu …
Zenazocine mesylate (USAN) 74559-85-6 Suppliers,provide Zenazocine mesylate (USAN) 74559-85-6 product and the products related with China (Mainland) Zenazocine mesylate (USAN) 74559-85-6 …
-
Zenazocine – Wikipedia
Zenazocine (INN; WIN-42,964) is an opioid analgesic of the benzomorphan family which made it to phase II clinical trials before development was ultimately halted and it was never marketed. [1][2] It …
-
Zenazocine | C23H35NO2 | ChemSpider
Structure, properties, spectra, suppliers and links for: Zenazocine, 74559-85-6, 77272-67-4.
-
Zenazocine mesylate (USAN) (CAS No. 74559-85-6) SDS
Zenazocine mesylate (USAN) CAS 74559-85-6 SDS including Hazard identification, Composition/information on ingredients, First-aid measures, Fire-fighting measures, Accidental …
-
Zenazocine – LookChem
Base Information Edit Chemical Name:Zenazocine CAS No.: 68681-43-6 Molecular Formula:C23H35NO2 Molecular Weight:357.534
-
74559-85-6 cas号74559-85-6分子式、结构式、MSDS、熔点、沸点
盖德化工字典提供Zenazocine mesylate (USAN)-CAS号74559-85-6百科,可查看CAS 74559-85-6分子量、密度、沸点、熔点、结构式、MSDS安全信息、生产方法及用途、核磁图谱等
-
68681-43-6 | Zenazocine | MOLCORE
MolCore offers CAS No.68681-43-6, Zenazocine for your process needs.Find product specific information including MFCD09838025,68681-43-6 MSDS,Price,Custom Synthesis.






Reviews
There are no reviews yet.