Nitric oxide (nitrogen oxide) Cas 10102-43-9
Nitric oxide (nitrogen oxide) Cas 10102-43-9
Nitric oxide (nitrogen oxide, nitrogen monooxide, or nitrogen monoxide[1]) is a colorless gas with the formula NO. It is one of the principal oxides of nitrogen. Nitric oxide is a free radical: it has an unpaired electron, which is sometimes denoted by a dot in its chemical formula (•N=O or •NO). Nitric oxide is also a heteronuclear diatomic molecule, a class of molecules whose study spawned early modern theories of chemical bonding.[6]
An important intermediate in industrial chemistry, nitric oxide forms in combustion systems and can be generated by lightning in thunderstorms. In mammals, including humans, nitric oxide is a signaling molecule in many physiological and pathological processes.[7] It was proclaimed the “Molecule of the Year” in 1992.[8] The 1998 Nobel Prize in Physiology or Medicine was awarded for discovering nitric oxide’s role as a cardiovascular signalling molecule.[9] Its impact extends beyond biology, with applications in medicine, such as the development of sildenafil (Viagra), and in industry, including semiconductor manufacturing. Nitric oxide (nitrogen oxide) Cas 10102-43-9
Nitric oxide should not be confused with nitrogen dioxide (NO2), a brown gas and major air pollutant, or with nitrous oxide (N2O), an anesthetic gas.[6]
History
Nitric oxide (NO) was first identified by Joseph Priestley in the late 18th century, originally seen as merely a toxic byproduct of combustion and an environmental pollutant.[12] Its biological significance was later uncovered in the 1980s when researchers Robert F. Furchgott, Louis J. Ignarro, and Ferid Murad discovered its critical role as a vasodilator in the cardiovascular system, a breakthrough that earned them the 1998 Nobel Prize in Physiology or Medicine. Nitric oxide (nitrogen oxide) Cas 10102-43-9
Physical properties
Electronic configuration
The ground-state electronic configuration of NO in united-atom notation is[14](1σ)2(2σ)2(3σ)2(4σ∗)2(5σ)2(1π)4(2π∗)1.The first two orbitals are actually pure atomic 1sO and 1sN from oxygen and nitrogen respectively and therefore are usually not noted in the united-atom notation. Orbitals noted with an asterisk are antibonding. The ordering of 5σ and 1π according to their binding energies is subject to discussion. Removal of a 1π electron leads to 6 states whose energies span over a range starting at a lower level than a 5σ electron an extending to a higher level. This is due to the different orbital momentum couplings between a 1π and a 2π electron. Nitric oxide (nitrogen oxide) Cas 10102-43-9
The lone electron in the 2π orbital makes NO a doublet (X 2Π) in its ground state, whose degeneracy is split in the fine structure from spin–orbit coupling with a total momentum J = 3/2 or J = 1/2. Nitric oxide (nitrogen oxide) Cas 10102-43-9
Dipole
The dipole of NO has been measured experimentally to 0.15740 D and is oriented from O to N (−NO+) due to the transfer of negative electronic charge from oxygen to nitrogen.[15]
Reactions
With di- and triatomic molecules
Upon condensing to a liquid, nitric oxide dimerizes to colorless dinitrogen dioxide (O=N–N=O), but the association is weak and reversible. The N–N distance in crystalline NO is 218 pm, nearly twice the N–O distance. Condensation in a highly polar environment instead gives the red alternant isomer O=N–O+=N−.[6]
Since the heat of formation of •NO is endothermic, NO can be decomposed to the elements. Catalytic converters in cars exploit this reaction: Nitric oxide (nitrogen oxide) Cas 10102-43-9
- 2 •NO → O2 + N2
When exposed to oxygen, nitric oxide converts into nitrogen dioxide:
- 2 •NO + O2 → 2 •NO2
This reaction is thought to occur via the intermediates ONOO• and the red compound ONOONO.[16]
In water, nitric oxide reacts with oxygen to form nitrous acid (HNO2). The reaction is thought to proceed via the following stoichiometry:
- 4 •NO + O2 + 2 H2O → 4 HNO2
Nitric oxide reacts with fluorine, chlorine, and bromine to form the nitrosyl halides, such as nitrosyl chloride:
- 2 •NO + Cl2 → 2 NOCl
With NO2, also a radical, NO combines to form the intensely blue dinitrogen trioxide:[6]
- •NO + •NO2 ⇌ ON−NO2
Organic chemistry
Nitric oxide rarely sees organic chemistry use. Most reactions with it produce complex mixtures of salts, separable only through careful recrystallization.[17]
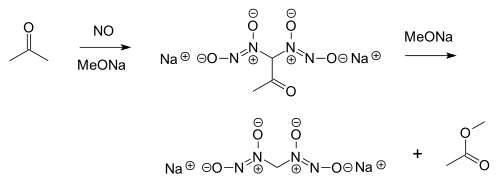
The addition of a nitric oxide moiety to another molecule is often referred to as nitrosylation. The Traube reaction is the addition of a two equivalents of nitric oxide onto an enolate, giving a diazeniumdiolate (also called a nitrosohydroxylamine).[18] The product can undergo a subsequent retro-aldol reaction, giving an overall process similar to the haloform reaction. For example, nitric oxide reacts with acetone and an alkoxide to form a diazeniumdiolate on each α position, with subsequent loss of methyl acetate as a by-product:[19]
This reaction, which was discovered around 1898, remains of interest in nitric oxide prodrug research. Nitric oxide can also react directly with sodium methoxide, ultimately forming sodium formate and nitrous oxide by way of an N-methoxydiazeniumdiolate.[20]
Sufficiently basic secondary amines undergo a Traube-like reaction to give NONOates.[21] However, very few nucleophiles undergo the Traube reaction, either failing to adduce NO or immediately decomposing with nitrous oxide release.[17]
Coordination complexes
Nitric oxide reacts with transition metals to give complexes called metal nitrosyls. The most common bonding mode of nitric oxide is the terminal linear type (M−NO).[6] Alternatively, nitric oxide can serve as a one-electron pseudohalide. In such complexes, the M−N−O group is characterized by an angle between 120° and 140°. The NO group can also bridge between metal centers through the nitrogen atom in a variety of geometries. Nitric oxide (nitrogen oxide) Cas 10102-43-9, Nitric oxide (nitrogen oxide) Cas 10102-43-9, Nitric oxide (nitrogen oxide) Cas 10102-43-9, Nitric oxide (nitrogen oxide) Cas 10102-43-9, Nitric oxide (nitrogen oxide) Cas 10102-43-9, Nitric oxide (nitrogen oxide) Cas 10102-43-9, Nitric oxide (nitrogen oxide) Cas 10102-43-9, Nitric oxide (nitrogen oxide) Cas 10102-43-9, Nitric oxide (nitrogen oxide) Cas 10102-43-9, Nitric oxide (nitrogen oxide) Cas 10102-43-9, Nitric oxide (nitrogen oxide) Cas 10102-43-9





Reviews
There are no reviews yet.