Buy 6-APDB (benzofuran) Cas 152623-93-3
Buy 6-APDB (benzofuran) Cas 152623-93-3
6-APDB, also known as 6-(2-aminopropyl)-2,3-dihydrobenzofuran or as 4-desoxy-MDA, is an entactogen of the phenethylamine, amphetamine, and dihydrobenzofuran families.[1] It is an analogue of MDA where the heterocyclic 4-position oxygen from the 3,4-methylenedioxy ring has been replaced with a methylene bridge.[1] 5-APDB (3-desoxy-MDA) is an analogue of 6-APDB where the 3-position oxygen has been replaced with a methylene instead.[1] 5-APDB was developed by a team led by David E. Nichols at Purdue University as part of their research into non-neurotoxic analogues of MDMA and first described in 1993.Buy 6-APDB (benzofuran) Cas 152623-93-3
Interactions
Pharmacology
Pharmacodynamics
In animal drug discrimination studies, 6-APDB fully substitutes for MBDB and MMAI but not for amphetamine or LSD.[1] In vitro, 6-APDB has been shown to inhibit the reuptake of serotonin, dopamine, and norepinephrine with IC50 values of 322 nM, 1,997 nM, and 980 nM, respectively.[1] These values are very similar to those of MDA, but with those for the catecholamines slightly lower in comparison, perhaps more similarly to MDMA.[1] Though 6-APDB does not substitute for amphetamine in rats at the doses used in referenced study, based on its in vitro profile it can be suggested that it may have amphetamine-like effects at higher doses. It also has activities at serotonin receptors.[7]
In subsequent animal studies, 6-APDB produced robust hyperlocomotion and, in drug discrimination tests, fully substituted for MDMA, partially substituted for DOM and cocaine, and failed to substitute for methamphetamine. Buy 6-APDB (benzofuran) Cas 152623-93-3
Chemistry
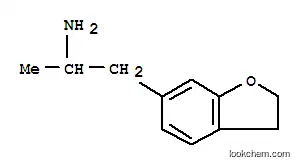
6-APDB, also known as 6-(2-aminopropyl)-2,3-dihydrobenzofuran, is a phenethylamine, amphetamine, and dihydrobenzofuran and an analogue of 3,4-methylenedioxyamphetamine (MDA). Buy 6-APDB (benzofuran) Cas 152623-93-3
Synthesis
The chemical synthesis of 6-APDB has been described.[6] Buy 6-APDB (benzofuran) Cas 152623-93-3
Analogues
In contrast to 6-APDB, 5-APDB is highly selective for serotonin.[1]
The unsaturated benzofuran derivative 6-APB, or 6-(2-aminopropyl)benzofuran is also known, but the difference in pharmacological effects between 6-APB and 6-APDB is unclear.
Buy 6-APDB (benzofuran) Cas 152623-93-3
CAS Number
Product Name
IUPAC Name
Molecular Formula
Molecular Weight
InChI
InChI Key
SMILES
Synonyms
Canonical SMILES
-
Synthesis and Characterization:
A 2011 study published in Microgram Journal details the synthesis, analysis, and characterization of 5- and 6-APDB []. This research provides details on the methods used to produce 6-APDB and techniques for its identification.
-
Classification and Effects:
6-APDB belongs to the class of phenethylamines and acts as a stimulant and entactogen []. Studies suggest it has similar effects to MDMA (3,4-methylenedioxymethamphetamine), but with potential differences in potency and specific mechanisms [].
6-(2-Aminopropyl)-2,3-dihydrobenzofuran, often referred to as 6-(2-aminopropyl)benzofuran, is a compound that belongs to the class of substituted benzofurans. This compound is structurally characterized by a benzofuran ring with a propylamine side chain at the sixth position. Its chemical formula is C11H15NO and it has a molecular weight of approximately 179.25 g/mol. The compound has garnered interest in both scientific research and recreational drug use due to its psychoactive properties.
- Oxidation: This compound can undergo oxidation reactions, which may lead to the formation of hydroxylated derivatives or other oxidized products.
- Reduction: It may also participate in reduction reactions, particularly when exposed to reducing agents.
- Rearrangement: Under specific conditions, this compound can undergo rearrangement reactions that alter its structure.
These reactions are essential for understanding the compound’s behavior in biological systems and its potential metabolic pathways .
The biological activity of 6-(2-aminopropyl)-2,3-dihydrobenzofuran is primarily linked to its effects on neurotransmitter systems. It acts as a serotonin-norepinephrine-dopamine reuptake inhibitor (SNDRI), which means it inhibits the reuptake of these neurotransmitters, potentially enhancing their levels in the synaptic cleft. This mechanism is similar to other psychoactive substances and contributes to its empathogenic effects . Additionally, it has been noted for its agonistic activity at serotonin receptors, particularly the 5-HT receptor subtype .
The synthesis of 6-(2-aminopropyl)-2,3-dihydrobenzofuran typically involves several steps:
- Starting Materials: The synthesis often begins with brominated phenols and bromoacetaldehyde diethylacetal.
- Formation of Benzofuran: The brominated phenol is treated with sodium hydride and heated to form a benzofuran structure.
- Amination: The resulting intermediate undergoes reductive amination with an appropriate amine to introduce the 2-aminopropyl group.
- Purification: The final product is purified through techniques such as column chromatography.
This multi-step synthesis highlights the complexity involved in producing this compound and ensures high purity for research purposes .
6-(2-Aminopropyl)-2,3-dihydrobenzofuran has various applications:
- Research Tool: It is used in pharmacological studies to understand its mechanism of action on neurotransmitter systems.
- Psychoactive Substance: Due to its empathogenic properties, it has been explored as a recreational drug.
- Potential Therapeutic Agent: Research is ongoing into its potential therapeutic applications in treating mood disorders or enhancing emotional processing.
Interaction studies involving 6-(2-aminopropyl)-2,3-dihydrobenzofuran focus on its effects on neurotransmitter transporters and receptors. These studies have shown that the compound interacts with serotonin transporter (SERT), norepinephrine transporter (NET), and dopamine transporter (DAT), indicating a complex pharmacological profile that may lead to both therapeutic benefits and risks associated with recreational use .
Several compounds share structural similarities with 6-(2-aminopropyl)-2,3-dihydrobenzofuran. Notable examples include:
These compounds highlight the uniqueness of 6-(2-aminopropyl)-2,3-dihydrobenzofuran due to its specific structural features and resultant pharmacological properties. Its empathogenic effects set it apart from others in its class. Buy 6-APDB (benzofuran) Cas 152623-93-3, Buy 6-APDB (benzofuran) Cas 152623-93-3, Buy 6-APDB (benzofuran) Cas 152623-93-3, Buy 6-APDB (benzofuran) Cas 152623-93-3, Buy 6-APDB (benzofuran) Cas 152623-93-3, Buy 6-APDB (benzofuran) Cas 152623-93-3, Buy 6-APDB (benzofuran) Cas 152623-93-3, Buy 6-APDB (benzofuran) Cas 152623-93-3, Buy 6-APDB (benzofuran) Cas 152623-93-3, Buy 6-APDB (benzofuran) Cas 152623-93-3, Buy 6-APDB (benzofuran) Cas 152623-93-3, Buy 6-APDB (benzofuran) Cas 152623-93-3, Buy 6-APDB (benzofuran) Cas 152623-93-3, Buy 6-APDB (benzofuran) Cas 152623-93-3
The synthesis of 6-(2-aminopropyl)-2,3-dihydrobenzofuran (6-APDB) originated in the early 1990s through the pioneering work of David E. Nichols at Purdue University. Nichols sought to develop analogs of 3,4-methylenedioxyamphetamine (MDA) and 3,4-methylenedioxymethamphetamine (MDMA) that retained entactogenic properties while minimizing neurotoxic side effects. His strategy involved structural modifications to the phenethylamine core, specifically replacing the methylenedioxy ring with a dihydrobenzofuran moiety. This substitution aimed to reduce oxidative stress associated with catecholamine metabolism, a key factor in MDMA-induced neurotoxicity.
6-APDB emerged alongside its positional isomer 5-APDB during systematic investigations into benzofuran-based phenethylamines. Initial in vitro studies revealed that 6-APDB inhibited serotonin (5-HT), dopamine (DA), and norepinephrine (NE) reuptake with IC~50~ values of 322 nM, 1,997 nM, and 980 nM, respectively. These values suggested a pharmacological profile intermediate between MDA (serotonin-preferring) and MDMA (balanced monoamine activity), positioning 6-APDB as a candidate for further behavioral characterization.
Nichols’ team employed drug discrimination paradigms in rats to assess subjective effects, finding that 6-APDB fully substituted for MBDB (N-methyl-1-(1,3-benzodioxol-5-yl)-2-butanamine) and MMAI (5-methoxy-6-methyl-2-aminoindane), but not for amphetamine or LSD. This specificity indicated a unique receptor interaction pattern distinct from classical stimulants or hallucinogens. Subsequent locomotor activity assays demonstrated 6-APDB-induced hyperlocomotion at doses comparable to MDMA, supporting its entactogen-like behavioral effects.
Table 1: Comparative Pharmacological Profiles of 6-APDB and Related Compounds
| Compound | SERT Inhibition (IC~50~, nM) | DAT Inhibition (IC~50~, nM) | NET Inhibition (IC~50~, nM) | 5-HT~2B~ Affinity (K~i~, nM) |
|---|---|---|---|---|
| 6-APDB | 322 | 1,997 | 980 | 3.7 |
| MDMA | 290 | 2,100 | 1,100 | 1,200 |
| MDA | 350 | 2,400 | 1,300 | 450 |
| 6-APB | 36 (release EC~50~) | 10 (release EC~50~) | 14 (release EC~50~) | 3.7 |
Chronological Evolution of Benzofuran-Based Phenethylamine Derivatives
The development of 6-APDB occurred within a broader context of structure-activity relationship (SAR) studies on benzofuran analogs. Following Nichols’ initial synthesis, researchers explored variations in ring saturation and substitution patterns:
- First-Generation Analogs (1990–2000):
- 5-APDB/6-APDB Pair: The dihydrobenzofuran scaffold allowed direct comparison of oxygen positioning. 5-APDB demonstrated serotonin-selective reuptake inhibition (SERT IC~50~ = 180 nM), while 6-APDB showed balanced monoamine activity.
- Benzofuran vs. Dihydrobenzofuran: Unsaturated derivatives like 6-APB (benzofuran) exhibited enhanced dopamine release (EC~50~ = 10 nM) compared to 6-APDB (EC~50~ = 1,997 nM).
- Second-Generation Modifications (2000–2010):
- N-Methylation: Introduction of N-methyl groups produced compounds like 5-MAPB and 6-MAPB. Surprisingly, N-methylation minimally affected transporter inhibition potency but increased 5-HT~2B~ receptor agonism (6-MAPB K~i~ = 2.8 nM).
- Ring Expansion: Tetrahydrobenzo[1,2-b:4,5-b’]difuran analogs explored conformational restriction, though these showed reduced entactogen activity.
- Third-Generation Hybrids (2010–Present):
- Benzofuran-Indole Chimeras: Combining benzofuran and tryptamine motifs yielded compounds with mixed 5-HT~2A~/DAT activity, though none surpassed 6-APDB’s balanced profile.
Table 2: Structural Evolution of Benzofuran Phenethylamines
| Generation | Key Modifications | Representative Compounds | Monoamine Selectivity Shift |
|---|---|---|---|
| 1st | Dihydrobenzofuran core | 5-APDB, 6-APDB | 5-HT > DA/NE → Balanced |
| 2nd | N-Alkylation, ring unsaturation | 6-APB, 5-MAPB | DA/NE > 5-HT |
| 3rd | Heterocyclic fusion | Benzodifuran analogs | 5-HT~2~ receptor selectivity |
Transition from Experimental Compound to Novel Psychoactive Substance (NPS)
6-APDB’s emergence in recreational markets followed a delayed trajectory compared to its analogs. Three phases characterized this transition:
- Academic to Gray Market (2005–2010):
- Patent analyses and forensic chemistry publications enabled small-scale synthesis by European research chemical vendors.
- Initial products appeared as “research chemicals” labeled for in vitro use, circumventing analog legislation.
- UK Proliferation (2011–2013):
- Marketed as “Benzofury” alongside 5-APB and 6-APB, 6-APDB gained popularity in club scenes due to its MDMA-like effects and legal status.
- Gas chromatography–mass spectrometry (GC-MS) analyses of seized samples confirmed 6-APDB’s presence in 23% of “Benzofury” products between 2011–2013.
- Regulatory Response (2013–Present):
- The UK Home Office classified 6-APDB as a Class B drug under the Misuse of Drugs Act 1971 in June 2013, citing structural similarity to MDA.
- Post-ban forensic prevalence decreased from 18% (2013) to <2% (2015) in European drug seizures, though darknet markets sustained limited availability.
6-(2-Aminopropyl)-2,3-dihydrobenzofuran exhibits potent inhibitory activity across all three major monoamine transporters, demonstrating a triple reuptake inhibition profile that underlies its psychoactive properties [3] [8]. In vitro studies utilizing rat brain synaptosomes have revealed that this compound functions as a substrate-type releaser rather than a simple reuptake inhibitor, distinguishing it from classical stimulants such as cocaine [8].
Serotonin Transporter Interactions
The compound demonstrates significant potency at the serotonin transporter with an inhibitory concentration fifty (IC50) value of 322 nanomolar [3]. This potency represents approximately 8.5-fold greater activity compared to 3,4-methylenedioxyamphetamine, indicating enhanced serotonergic activity [8]. In release assays conducted in rat brain synaptosomes, 6-(2-Aminopropyl)-2,3-dihydrobenzofuran produced concentration-dependent release of [³H]5-hydroxytryptamine with an effective concentration fifty (EC50) of 36 nanomolar, achieving 100% efficacy relative to maximal release [8].
Dopamine Transporter Dynamics
At the dopamine transporter, 6-(2-Aminopropyl)-2,3-dihydrobenzofuran exhibits an IC50 value of 1,997 nanomolar for uptake inhibition, demonstrating preferential activity for serotonergic over dopaminergic systems [3]. However, the compound functions as a potent dopamine releaser with an EC50 of 10 nanomolar, representing 10-fold greater potency than 3,4-methylenedioxyamphetamine [8]. This substrate-type releasing activity at the dopamine transporter involves reversal of normal transporter flux, consistent with the molecular mechanism observed for ring-substituted amphetamines [8].
Norepinephrine Transporter Modulation
The norepinephrine transporter represents an intermediate target for 6-(2-Aminopropyl)-2,3-dihydrobenzofuran, with an IC50 value of 980 nanomolar for uptake inhibition [3]. Release studies demonstrate an EC50 of 14 nanomolar for norepinephrine efflux, indicating potent substrate activity at this transporter [8]. The compound achieves 98% efficacy relative to maximal norepinephrine release, suggesting full substrate properties at the norepinephrine transporter [8].
Comparative Transporter Activity Profile
| Transporter System | IC50 Inhibition (nM) | EC50 Release (nM) | Relative Potency vs MDA |
|---|---|---|---|
| Serotonin Transporter | 322 | 36 | 4.5-fold more potent |
| Dopamine Transporter | 1,997 | 10 | 10-fold more potent |
| Norepinephrine Transporter | 980 | 14 | 3.4-fold more potent |
The transporter selectivity profile indicates non-selective substrate activity across all three monoamine transporters, with calculated ratios suggesting balanced effects on multiple neurotransmitter systems [8]. This pharmacological profile distinguishes 6-(2-Aminopropyl)-2,3-dihydrobenzofuran from selective serotonin reuptake inhibitors and demonstrates its classification as a broad-spectrum monoamine releaser [8].
Serotonergic Receptor Agonism: 5-Hydroxytryptamine2A/2B/2C Binding Affinity Landscapes
6-(2-Aminopropyl)-2,3-dihydrobenzofuran demonstrates significant agonist activity at multiple serotonin receptor subtypes, with particularly pronounced effects at the 5-hydroxytryptamine2B receptor [12] [16]. These receptor interactions contribute to the compound’s complex pharmacological profile and distinguish it from pure transporter inhibitors
152623-93-3
-
-
Basic information
- Product Name: 6-(2-aminopropyl)-2,3-dihydrobenzofuran
- Synonyms: 6-(2-aminopropyl)-2,3-dihydrobenzofuran;6-APDB;6-BenzofuranethanaMine,2,3-dihydro-a-Methyl-;1-(2,3-dihydrobenzofuran-6-yl)propan-2-amine;6-APDB lila@tuskwei.com whatsapp:+8615230133735
- CAS NO:152623-93-3
- Molecular Formula: C11H15NO
- Molecular Weight: 177.11
- EINECS: N/A
- Product Categories: pharmaceutical intermediate
- Mol File: 152623-93-3.mol
-
Chemical Properties
- Melting Point: N/A
- Boiling Point: 292.868 °C at 760 mmHg
- Flash Point: 134.233 °C
- Appearance: /
- Density: 1.088 g/cm3
- Vapor Pressure: 0.00179mmHg at 25°C
- Refractive Index: 1.565
- Storage Temp.: N/A
- Solubility: N/A
- CAS DataBase Reference: 6-(2-aminopropyl)-2,3-dihydrobenzofuran(CAS DataBase Reference)
- NIST Chemistry Reference: 6-(2-aminopropyl)-2,3-dihydrobenzofuran(152623-93-3)
- EPA Substance Registry System: 6-(2-aminopropyl)-2,3-dihydrobenzofuran(152623-93-3)
-
Safety Data
- Hazard Codes: N/A
- Statements: N/A
- Safety Statements: N/A
- WGK Germany:
- RTECS:
- HazardClass: N/A
- PackingGroup: N/A
- Hazardous Substances Data: 152623-93-3(Hazardous Substances Data)
152623-93-3 Suppliers
Recommended suppliers
152623-93-3 Usage
Uses
Used in Recreational Drug Industry:
6-(2-aminopropyl)-2,3-dihydrobenzofuran is used as a recreational drug for its psychoactive effects, which include euphoria, hallucinations, and altered perception. It is particularly sought after by individuals who are interested in experiencing these effects for personal reasons.
Used in Research Chemical Industry:
6-APDB is also used as a research chemical, where it may be studied for its potential effects and interactions within the scientific community. Researchers may explore its properties to better understand its mechanisms of action and potential applications in various fields.
Check Digit Verification of cas no
The CAS Registry Mumber 152623-93-3 includes 9 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 6 digits, 1,5,2,6,2 and 3 respectively; the second part has 2 digits, 9 and 3 respectively.
Calculate Digit Verification of CAS Registry Number 152623-93:
(8*1)+(7*5)+(6*2)+(5*6)+(4*2)+(3*3)+(2*9)+(1*3)=123
123 % 10 = 3
So 152623-93-3 is a valid CAS Registry Number.
152623-93-3SDS
SAFETY DATA SHEETS
According to Globally Harmonized System of Classification and Labelling of Chemicals (GHS) – Sixth revised edition
Version: 1.0
Creation Date: Aug 20, 2017
Revision Date: Aug 20, 2017
1.Identification
1.1 GHS Product identifier
| Product name | 1-(2,3-dihydro-1-benzofuran-6-yl)propan-2-amine |
|---|
1.2 Other means of identification
| Product number | – |
|---|---|
| Other names | 6-Nprop-dbf |
1.3 Recommended use of the chemical and restrictions on use
| Identified uses | For industry use only. |
|---|---|
| Uses advised against | no data available |
1.4 Supplier’s details
1.5 Emergency phone number
| Emergency phone number | – |
|---|---|
| Service hours | Monday to Friday, 9am-5pm (Standard time zone: UTC/GMT +8 hours). |
More Details:152623-93-3 SDS
152623-93-3Upstream product
- 152623-98-8N-(trifluoroacetyl)-1-<3-methoxy-4-(chloroacetyl)phenyl>-2-aminopropane
- 152623-99-96-<2-<n-(trifluoroacetyl)amino>propyl>-2,3-dihydrobenzofuran-3-one </n-(trifluoroacetyl)amino>
- 152623-97-7N-(trifluoroacetyl)-1-(3-methoxyphenyl)-2-aminopropane
- 17862-85-03-methoxyamphetamine
- 152624-00-56-<2-<n-(trifluoroacetyl)amino>propyl>-2,3-dihydrobenzofuran </n-(trifluoroacetyl)amino>
152623-93-3Downstream Products
152623-93-3Relevant academic research and scientific papers
Successful use of a novel lux i-Amylose-1 chiral column for enantioseparation of “legal highs” by HPLC
Kadkhodaei, Kian,Kadisch, Marlene,Schmid, Martin G.
, p. 42 – 52 (2019/11/14)
Bath salts, fumigations, cleaners and air fresheners, behind these terms substances are hidden, which count as “Legal Highs”. These fancy names are used to pretend Legal Highs as harmless compounds, to circumvent legal regulations for marketing as well as to increase the sales. Besides classic illicit drugs of synthetic origin such as amphetamines, cocaine and MDMA, the trade of these compounds, also known as new psychoactive substances (NPS), is not uncommon today. In many countries, NPS are still not subject to drug control. Among them, there are stimulants such as new amphetamine derivatives or cathinones, which possess a chiral centre. Little is known about the fact that the two possible enantiomers may differ in their pharmacological effect. The aim of this study was to test a novel HPLC column for the enantioseparation of a set of 112 NPS coming from different chemical groups and collected by internet purchases during the years 2010–2018. The CSP, namely Lux 5?μm i-Amylose-1, LC Column 250 x 4.6?mm, was run in normal phase mode under isocratic conditions, UV detection was performed at 245?nm and 230?nm, injection volume was 10?μl and flow rate was 1?ml/min. With a mobile phase consisting of n-hexane/isopropanol/diethylamine (90:10:0.1), herein, 79 NPS were resolved into their enantiomers successfully, for 37 of them baseline resolution was achieved. After increase of lipophily of the mobile phase to 99:1:0.1, another 27 compounds were baseline separated. It was found that all separated NPS are traded as racemic compounds.
Enantiomeric separation of Novel Psychoactive Substances by capillary electrophoresis using (+)-18-crown-6-tetracarboxylic acid as chiral selector
H?gele, Johannes S.,Schmid, Martin G.
, p. 1019 – 1026 (2018/07/29)
In the recent years, hundreds of Novel Psychoactive Substances (NPS) have entered both the European and the global drug market. These drugs, which are mainly used for recreational matters, have caused serious social problems. Every year, the spectrum of these misused drugs is enlarged by new derivatives, which are produced by modifications of basic structures of already well-known substances. Additionally, a lot of them possess a stereogenic center which leads to 2 enantiomeric forms. The fact that the pharmacological effects and potencies of the enantiomers of these chiral NPS may differ can be assumed from a broad spectrum of active pharmaceutical ingredients. For this reason, analytical method development regarding enantiomeric separation for these classes of substances is of great pharmaceutical and medical interest. The aim of this work was to create an easy-to-prepare chiral capillary electrophoresis method for the enantioseparation of NPS which contains a primary amino group by means of (+)-18-crown-6-tetracarboxylic acid as chiral selector. Novel Psychoactive Substances were purchased at various Internet stores or represent samples seized by Austrian police. The effects of selector concentration, the electrolyte composition, and the addition of organic modifiers to the background electrolyte on enantioseparation were investigated. Under optimized conditions, the use of 20-mM (+)-18-crown-6-tetracarboxylic acid, 10-mM Tris, and 30-mM citric acid buffer at pH 2.10 turned out to be effective. Fifteen of 24 tested NPS were resolved in their enantiomers within 15?minutes. It was found that all NPS were traded as racemic mixtures.
Enantioselective potential of polysaccharide-based chiral stationary phases in supercritical fluid chromatography
Kucerova, Gabriela,Kalikova, Kveta,Tesarova, Eva
supporting information, p. 239 – 246 (2017/05/29)
The enantioselective potential of two polysaccharide-based chiral stationary phases for analysis of chiral structurally diverse biologically active compounds was evaluated in supercritical fluid chromatography using a set of 52 analytes. The chiral selectors immobilized on 2.5?μm silica particles were tris-(3,5-dimethylphenylcarmabate) derivatives of cellulose or amylose. The influence of the polysaccharide backbone, different organic modifiers, and different mobile phase additives on retention and enantioseparation was monitored. Conditions for fast baseline enantioseparation were found for the majority of the compounds. The success rate of baseline and partial enantioseparation with cellulose-based chiral stationary phase was 51.9% and 15.4%, respectively. Using amylose-based chiral stationary phase we obtained 76.9% of baseline enantioseparations and 9.6% of partial enantioseparations of the tested compounds. The best results on cellulose-based chiral stationary phase were achieved particularly with propane-2-ol and a mixture of isopropylamine and trifluoroacetic acid as organic modifier and additive to CO2, respectively. Methanol and basic additive isopropylamine were preferred on amylose-based chiral stationary phase. The complementary enantioselectivity of the cellulose- and amylose-based chiral stationary phases allows separation of the majority of the tested structurally different compounds. Separation systems were found to be directly applicable for analyses of biologically active compounds of interest.
Synthesis and Pharmacological Examination of Benzofuran, Indan, and Tetralin Analogues of 3,4-(Methylenedioxy)amphetamine
Monte, Aaron P.,Marona-Lewicka, Danuta,Cozzi, Nicholas V.,Nichols, David E.
, p. 3700 – 3706 (2007/10/02)
Benzofuran, indan and tetrahydronaphthalene analogs of 3,4-(methylenedioxy)amphetamine (MDA) were prepared in order to examine the role of the dioxole ring oxygen atoms of MDA in interacting with the serotonin and catecholamine uptake carriers.The series of compounds was evaluated for discriminative stimulus effects in rats trained to discriminate saline from the training drugs (S)-(+)-MBDB (1c), MMAI (3), and (S)-(+)-amphetamine and for the ability to inhibit the uptake of 3H>serotonin, 3H>dopamine, and 3H>norepinephrine into crude synaptosome preparations.Behaviorally, the benzofuran and indan analogs 4-6 produced similar discriminative cues, whereas the tetralin derivative 7 did not fully substitute for the training drugs.The results in the in vitro pharmacology studies indicate that selectivity for 5-HT versus catecholamine uptake carriers may be modulated by the position and orientation of ring oxygen atoms.However, the nonoxygenated isostere 6 possessed high potency at all uptake sites examined.Enlargement of the saturated ring by one methylene unit to give the tetralin derivative resulted in a large (3-4-fold) reduction in activity at catecholamine sites.
5-Hydroxytryptamine2B Receptor Interactions
-
6- (2-aminopropyl)-2,3-dihydrobenzofuran | 152623-93-3 – ChemicalBook
Apr 29, 2025 · 6- (2-aminopropyl)-2,3-dihydrobenzofuran (CAS 152623-93-3) information, including chemical properties, structure, melting point, boiling point, density, formula, molecular weight, uses, …
-
6-APDB CAS # 152623-93-3 – bismanpharma.com
6-APDB CAS # 152623-93-3 or 6- (2-Aminopropyl)-2,3-dihydrobenzofuran (6-APDB, 4-Desoxy-MDA, EMA-3) is a stimulant and entactogen drug of the phenethylamine and amphetamine classes. 6 …
-
6-APDB 152623-93-3 Purity 99.99% discretely and vacuum sealed …
Cas NO.: 152623-93-3 Name: 6- (2-aminopropyl)-2,3-dihydrobenzofuran Molecular Formula: C11H15NO Boiling Point: 292.9°Cat760mmHg Refractive index: 1.565 Flash Point: 134.2°C Purity: 99.99% …
-
Cas 152623-93-3,6- (2-aminopropyl)-2,3-dihydrobenzofuran | lookchem
It consists of a benzene ring fused with a furan ring and a 2-aminopropyl side chain. 6- (2-aminopropyl)-2,3-dihydrobenzofuran is also known as 6-APDB and is classified as a designer drug or research …
-
1- (2,3-Dihydro-1-benzofuran-6-yl)-2-propanamine (CAS 152623-93-3) …
1- (2,3-Dihydro-1-benzofuran-6-yl)-2-propanamine (CAS: 152623-93-3) – Find 5 suppliers offering this chemical compound. Access comprehensive data: molecular formula C11H15NO, molecular weight …
-
6-APDB – Chemical Route
6-APDB is a research chemical that is commonly used as a stimulant and an entactogenic compound. It is a relative analogue of 6-APB, a cyclic analogue of the phenylalanine-MDA and can be commonly …
-
Buy 6- (2-Aminopropyl)-2,3-dihydrobenzofuran | 152623-93-3
Buy 6- (2-Aminopropyl)-2,3-dihydrobenzofuran, CAS No. 152623-93-3 from Smolecule. Molecular Formula C11H15NO. Molecular Weight 177.24 g/mol.
-
Buy 6-(2-aminopropyl)-2,3-dihydrobenzofuran from LEAP CHEM CO., …
LEAP CHEM CO., LTD., with approximately 1613890 visits to its shop, is a reputable Trader that has been providing 6- (2-aminopropyl)-2,3-dihydrobenzofuran products for 3 years on ECHEMI.
-
CAS 152623-93-3 6-Benzofuranethanamine,2,3-dihydro-a-methyl
Benzofuranamine derivative for CNS-active compound research. Provides bioactive scaffold for pharmaceutical discovery.
-
1- (2,3-DIHYDRO-1-BENZOFURAN-6-YL)PROPAN-2-AMINE
CAS:152623-93-3 1- (2,3-dihydro-1-benzofuran-6-yl)propan-2-amine manufacturer supplier,contact us for PRICE,IR,NMR,GC,HPLC Chart,COA,MSDS or Route of Synthesis



Reviews
There are no reviews yet.