Buy Salvinorin A Cas 83729-01-5
Buy Salvinorin A Cas 83729-01-5
Salvinorin A is the main active psychotropic molecule in Salvia divinorum. Salvinorin A is considered an atypical dissociative hallucinogen.[3][4][5]
It is structurally distinct from other naturally occurring hallucinogens (such as DMT, psilocybin, and mescaline) because it contains no nitrogen atoms; hence, it is not an alkaloid (and cannot be rendered as a salt), but rather is a terpenoid.[4] It also differs in subjective experience, compared to other hallucinogens, and has been described as having strong dissociative effects. Buy Salvinorin A Cas 83729-01-5
Salvinorin A can produce psychoactive experiences in humans with a typical duration of action being several minutes to an hour or so, depending on the method of ingestion.[6]
Salvinorin A is found with several other structurally related salvinorins. Salvinorin is a trans–neoclerodane diterpenoid. It acts as a kappa opioid receptor agonist and is the first known compound acting on this receptor that is not an alkaloid.[6]
History
Salvinorin A was first described and named in 1982 by Alfredo Ortega and colleagues in Mexico. They used a combination of spectroscopy and x-ray crystallography to determine the chemical structure of the compound, which was shown to have a bicyclic diterpene structure.[7] Around the same time, Leander Julián Valdés III independently isolated the molecule as part of his PhD research, published in 1983.[8] Valdés named the chemical divinorin, and also isolated an analog that he named divinorin B. The naming was subsequently corrected to salvinorin A and B after the work was published in 1984.[9] Valdés later isolated salvinorin C. Buy Salvinorin A Cas 83729-01-5
Pharmacology
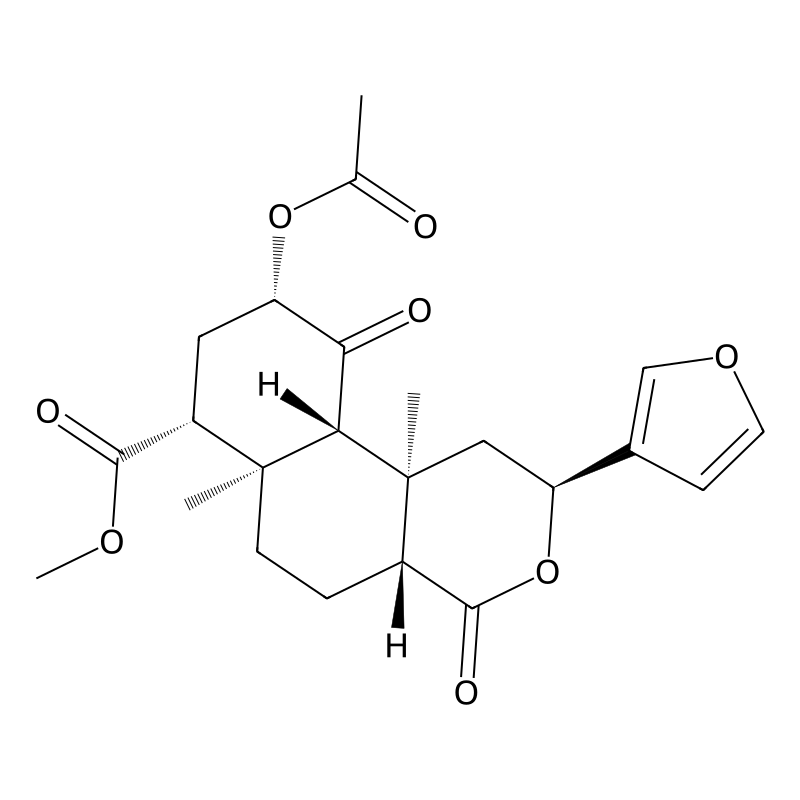
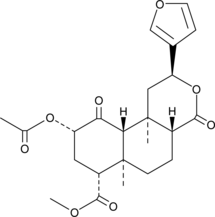
Salvinorin A is a trans-neoclerodane diterpenoid with the chemical formula C23H28O8.[11] Unlike other known opioid-receptor ligands, salvinorin A is not an alkaloid, as it does not contain a nitrogen atom.[4][12] Salvinorin A has no action at the 5-HT2A serotonin receptor, the principal molecular target responsible for the actions of ‘classical’ psychedelics such as LSD and mescaline.[6][12] Salvinorin A has also been shown to have effect on cannabinoid CB1 receptors.[13] It significantly increases prolactin and inconsistently increases cortisol.[14] It causes dysphoria by stopping release of dopamine in the striatum.[15] Salvinorin A increases activity of DAT while decreasing activity of SERT.[15]
Pharmacokinetics
Salvinorin A is effectively deactivated by the gastrointestinal system, so alternative routes of administration must be used for better absorption. It is absorbed by oral mucosa.[16] It has a half-life of around 8 minutes in non-human primates.[17]
Potency and selectivity
Salvinorin A is active at doses as low as 200 μg.[11][18][19] Synthetic chemicals, such as LSD (active at 20–30 μg doses), can be more potent.[20] Research has shown that salvinorin A is a potent κ-opioid receptor (KOR) agonist (Ki = 2.4 nM, EC50 = 1.8 nM).[11] It has a high affinity for the receptor, indicated by the low dissociation constant of 1.0 nanomolar (nM).[21] In addition, salvinorin A has been found to act as a D2 receptor partial agonist, with an affinity of 5–10 nM, an intrinsic activity of 40–60%, and an EC50 of 48 nM.[22] This suggests that the D2 receptor may also play an important role in its effects.[22]
Salvinorin A shows atypical properties as an agonist of the KOR relative to other KOR agonists.[23]
Effect on intestinal motility
Salvinorin A is capable of inhibiting excess intestinal motility (e.g. diarrhea), through its potent κ-opioid-activating effects. The mechanism of action for salvinorin A on ileal tissue has been described as ‘prejunctional’, as it was able to modify electrically induced contractions, but not those of exogenous acetylcholine.[24] A pharmacologically important aspect of the contraction-reducing properties of ingested salvinorin A on gut tissue is that it is only pharmacologically active on inflamed and not normal tissue, thus reducing possible side-effects. Buy Salvinorin A Cas 83729-01-5
Solubility
Salvinorin A is soluble in organic solvents such as ethanol and acetone, but not especially so in water.[26]
Detection in urine
Researchers found that humans who smoked 580 μg of the pure drug had urine salvinorin A concentrations of 2.4–10.9 μg/L during the first hour; the levels fell below the detection limit by 1.5 hours after smoking.[27]
Research
Salvinorin A has only been administered to humans in a few studies, one showing that its effects peaked at about 2 minutes, that its subjective effects may overlap with those of serotonergic psychedelics, and that it temporarily impairs recall and recognition memory.[5] Like most other agonists of KOR, salvinorin A produces sedation, psychotomimesis, dysphoria, anhedonia, and depression.[4][28][29] Salvinorin A has been screened for its possible use as a structural “scaffold” in medicinal chemistry in developing new drugs for treating psychiatric diseases[4][30] such as cocaine dependence.[31]
Synthesis

Biosynthesis
The biogenic origin of salvinorin A synthesis has been elucidated using nuclear magnetic resonance and ESI–MS analysis of incorporated precursors labeled with stable isotopes of carbon (carbon-13 13C) and hydrogen (deuterium 2H). It “is biosynthesized via the 1-deoxy-d-xylulose-5-phosphate pathway“, rather than the classic mevalonate pathway, consistent with the common plastidial localization of diterpenoid metabolism.
Salvinorin A
Content Navigation
CAS Number
Product Name
IUPAC Name
Molecular Formula
Molecular Weight
InChI
InChI Key
SMILES
solubility
Synonyms
Canonical SMILES
Isomeric SMILES
Selective Kappa Opioid Receptor Agonism
Salvinorin A’s primary area of scientific interest lies in its potent and selective binding to kappa opioid receptors (KORs) in the brain. Unlike traditional opioid drugs that target mu (µ) opioid receptors, SA acts specifically on KORs, producing distinct pharmacological effects []. This selectivity is crucial as mu-opioid receptor activation is associated with pain relief but also carries the risk of dependence and respiratory depression.
Source
Potential Therapeutic Applications
Preclinical studies in animal models suggest several promising therapeutic applications for Salvinorin A:
- Antidepressant Effects: Research suggests SA may have antidepressant properties. Studies have shown it can rapidly reverse social withdrawal behavior in mice, a symptom of depression [].
- Addiction Treatment: Due to its unique interaction with KORs, SA might hold promise for treating addiction. Studies indicate it may reduce the rewarding effects of addictive drugs like cocaine [].
- Neuroprotective Properties: Salvinorin A exhibits neuroprotective effects in animal models of stroke and brain injury. It may help protect brain cells from damage caused by oxygen deprivation.
Salvinorin A is a potent psychoactive compound primarily found in the plant Salvia divinorum, a member of the mint family native to Mexico. It is classified as a neoclerodane diterpenoid with the chemical formula C23H28O8. Distinctively, salvinorin A is recognized as the most potent naturally occurring hallucinogen, possessing unique properties that differentiate it from traditional hallucinogens such as lysergic acid diethylamide and psilocybin, which primarily act on serotonin receptors. Instead, salvinorin A selectively agonizes kappa opioid receptors in the brain, leading to its characteristic effects of intense dissociation and altered perception .
Salvinorin A
Item No. 11487
Synonyms
- Divinorin A
- Sal A
Technical Information
Shipping & Storage Information
- Unlike classical hallucinogens that target serotonin receptors, Salvinorin A acts as a potent and selective agonist at kappa-opioid receptors (KORs) in the central nervous system.
- KOR activation by Salvinorin A is believed to be responsible for its dissociative hallucinogenic effects, causing distortions in perception, detachment from reality, and intense emotional experiences.
- Salvinorin A is a highly potent hallucinogen with a rapid onset and short duration of action (minutes).
- Due to its intense effects, it can lead to psychological distress, anxiety, paranoia, and difficulty controlling movement.
- There is limited data on toxicity, but high doses can cause respiratory depression and seizures.
- Salvinorin A is a Schedule I controlled substance in many countries due to its high potential for abuse and lack of accepted medical use.
The biosynthesis of salvinorin A involves several key chemical transformations, including oxygenation, acylation, and methylation reactions. It is synthesized from two five-carbon precursors: isopentenyl diphosphate and dimethylallyl diphosphate, through the 1-deoxy-D-xylulose 5-phosphate pathway. The initial reaction is catalyzed by ent-clerodienyl diphosphate synthase, which produces a carbocation that rearranges to form the clerodane scaffold characteristic of salvinorin A .
Salvinorin A’s chemical structure includes multiple functional groups that contribute to its stability and reactivity. The compound has a high melting point (238–240 °C) and is poorly soluble in water, complicating its formulation for therapeutic use .
Salvinorin A exhibits a range of biological activities primarily through its action as a selective kappa opioid receptor agonist. This interaction leads to various effects including:
- Dissociation: Users often report a profound disconnection from reality.
- Hallucinations: Intense visual and auditory experiences are common.
- Neuroprotective Effects: Research indicates potential applications in treating conditions related to hypoxia or ischemia due to its neuroprotective properties .
In animal studies, salvinorin A has shown promise in addressing addiction and depression, suggesting potential therapeutic applications beyond recreational use .
Salvinorin A has garnered interest for various applications:
- Research Tool: Its unique mechanism of action makes it valuable in studying kappa opioid receptor functions.
- Therapeutic Potential: Emerging studies suggest possible uses in treating psychiatric disorders such as depression and anxiety due to its neuroprotective and anti-addiction properties .
- Psychoactive Use: Recreationally, it is used for its intense psychoactive effects, typically administered through smoking or chewing the leaves of Salvia divinorum .
Research on salvinorin A has focused on its interactions with various receptors:
- Kappa Opioid Receptors: Salvinorin A’s primary action is through kappa opioid receptors, leading to analgesic and sedative effects .
- Serotonin Receptors: Unlike classical psychedelics that target serotonin receptors (5HT2A), salvinorin A does not exhibit affinity for these sites, marking a significant distinction in its pharmacological profile .
- Dopamine Modulation: Salvinorin A has been shown to inhibit dopamine release in certain brain regions, contributing to its dysphoric effects at higher doses .
Several compounds are structurally similar to salvinorin A but differ significantly in their psychoactive properties:
| Compound Name | Structure Type | Psychoactive Properties | Notes |
|---|---|---|---|
| Salvinorin B | Neoclerodane Diterpenoid | Low psychoactivity | Hydrolysis product of salvinorin A |
| Enadoline | Kappa Opioid Agonist | Moderate psychoactivity | Older compound with unpleasant effects |
| Dynorphin | Peptide | Analgesic but not hallucinogenic | Endogenous peptide affecting opioid receptors |
| U50,488 | Synthetic Kappa Agonist | Moderate psychoactivity | Used in research but less potent than salvinorin A |
Salvinorin A stands out due to its extraordinary potency and rapid onset of effects compared to these other compounds. Its unique mechanism of action at kappa opioid receptors contributes significantly to its distinct psychotropic profile.
Feedback inhibition mechanisms play crucial roles in pathway regulation, with the end products isopentenyl diphosphate and dimethylallyl diphosphate exhibiting inhibitory effects on deoxyxylulose 5-phosphate synthase [6]. This regulatory mechanism ensures tight control of carbon flux through the pathway and prevents overaccumulation of isoprenoid precursors [6].
Enzymatic Modifications and Secondary Metabolism
The transformation of geranylgeranyl diphosphate into Salvinorin A involves a series of specialized enzymatic modifications catalyzed by diterpene synthases and cytochrome P450 enzymes [9] [10] [15]. The first committed step in Salvinorin A biosynthesis is mediated by the class II diterpene synthase SdCPS2, also known as clerodienyl diphosphate synthase [16] [17].
SdCPS2 catalyzes the protonation-initiated cyclization of geranylgeranyl diphosphate through the formation of the common bicyclic intermediate labda-13-en-8-yl diphosphate [9] [10]. This intermediate undergoes a series of Wagner-Meerwein rearrangements involving two sequential 1,2-hydride and methyl shifts that result in the formation of the neoclerodane scaffold characteristic of Salvinorin A [18]. The reaction is terminated by proton abstraction to yield kolavenyl diphosphate, which retains the diphosphate group from the original substrate [9] [10].
Alternative nomenclature in the literature refers to this enzyme as SdKPS or kolavenyl diphosphate synthase, reflecting its specific product formation [9] [19]. The enzyme exhibits remarkable stereochemical control, producing exclusively the negative enantiomer of kolavenyl diphosphate through precise positioning of catalytic residues within the active site [9].
The subsequent dephosphorylation of kolavenyl diphosphate to form kolavenol represents a critical step in the pathway that has been the subject of considerable investigation [15] [20]. While initially proposed to be catalyzed by a class I diterpene synthase, more recent evidence suggests that this conversion may be mediated by a phosphatase activity rather than a traditional class I diterpene synthase [15].
Following the formation of kolavenol, the biosynthetic pathway diverges into multiple oxidative modifications catalyzed by cytochrome P450 enzymes [21] [22]. Recent research has identified three specific cytochrome P450 enzymes responsible for the sequential conversion of kolavenol into the immediate precursors of Salvinorin A [21].
The first cytochrome P450-catalyzed transformation involves the conversion of kolavenol to annonene by the enzyme designated as annonene synthase [21]. This enzyme introduces specific oxidative modifications that establish the foundation for subsequent furan ring formation characteristic of neoclerodane diterpenoids [21].
The second oxidative step is catalyzed by hardwickiic acid synthase, which converts annonene to hardwickiic acid [21]. Hardwickiic acid represents a crucial intermediate that has been isolated from Salvia divinorum and exhibits pharmacological activity at kappa-opioid receptors, albeit with lower potency compared to Salvinorin A [23].
The formation of crotonolide G from kolavenol is catalyzed by the cytochrome P450 enzyme SdCS, which has been characterized as CYP76AH39 [15] [20]. This enzyme incorporates molecular oxygen through a mechanism involving hydroxylation at the C16 position followed by nucleophilic attack by the hydroxyl group at C15, resulting in dihydrofuran ring formation [20].
| Table 2: Enzymatic Modifications in Salvinorin A Biosynthesis |
|---|
-
Salvinorin A – MilliporeSigma
Salvinorin A; CAS Number: 83729-01-5; Synonyms: Salvinorin A,κ-opioid Receptor Agonist, Salvinorin A, Divinorin A at Sigma-Aldrich
-
Salvinorin A (Divinorin A), kappa agonist (CAS 83729 …
MW 432.5 Da, Purity >98%. Potent κ agonist (K i = 8-19 nM). Psychoactive and antinociceptive in vivo. Reported to be an allosteric modulator of μ opioid …
-
Buy Salvinorin A | 83729-01-5 | >98% – smolecule.com
Aug 15, 2023 · Description Salvinorin A is a potent psychoactive compound primarily found in the plant Salvia divinorum, a member of the mint family native …
-
Salvinorin A (CAS 83729-01-5) – Cayman Chemical
Salvinorin A: An Analytical Reference Material. CAS Number: 83729-01-5. Synonyms: Divinorin A, Sal A. Purity: ≥95%.
-
Salvinorin A | CAS 83729-01-5 | AbMole BioScience
Salvinorin A is a potent, non-nitrogenous κ opioid selective receptor agonist. Buy …
-
CAS Number: 83729-01-5
-
Molecular Weight: 432.46
-
Formula: C23H28O8
-
Solubility (25°C): DMSO: ≥10 mg/mL
-
-
SALVINORIN A | 83729-01-5 – ChemicalBook
Jan 13, 2026 · SALVINORIN A (CAS 83729-01-5) information, including chemical properties, structure, melting point, boiling point, density, formula, molecular weight, uses, prices, suppliers, SDS and …
-
CAS No : 83729-01-5 | Chemical Name : SALVINORIN A
Buy highly pure SALVINORIN A, CAS No : 83729-01-5, Mol.Formula : C23H28O8, Mol.Weight : 432.5, from Pharmaffiliates. Login as registered user for prices, availability and discounts.
Salvinorin A | CAS No. 83729-01-5 | Clearsynth
Buy Salvinorin A CAS No- 83729-01-5, a high quality product from Clearsynth, a Leading Manufacturer and Exporter of Salvinorin A accompanied by Certificate of Analysis.
-
Salvinorin A – CAS 83729-01-5 – Calbiochem | 508159 – Merck
Salvinorin A – CAS 83729-01-5 – Calbiochem – Find MSDS or SDS, a COA, data sheets and more information.
-
83729-01-5 | Salvinorin A | ChemScene
ChemScene Provide CAS 83729-01-5, Salvinorin A, Divinorin A, Formula:C23H28O8, MW:432.4600. We also provide services including custom synthesis, process optimization, commercial production, …




Reviews
There are no reviews yet.